BACKGROUND
Evidence to support the choice of blood-pressure targets for the treatment of coma- tose survivors of out-of-hospital cardiac arrest who are receiving intensive care is limited.
对于正在接受重症监护的院外心脏停搏后昏迷幸存者,为血压目标选择提供支持的证据有限。
METHODS
In a double-blind, randomized trial with a 2-by-2 factorial design, we evaluated a mean arterial blood-pressure target of 63 mm Hg as compared with 77 mm Hg in coma-tose adults who had been resuscitated after an out-of-hospital cardiac arrest of presumed cardiac cause; patients were also assigned to one of two oxygen targets (reported separately). The primary outcome was a composite of death from any cause or hospital discharge with a Cerebral Performance Category (CPC) of 3 or 4 within 90 days (range, 0 to 5, with higher categories indicating more severe disability; a category of 3 or 4 indicates severe disability or coma). Secondary outcomes included neuron-specific enolase levels at 48 hours, death from any cause, scores on the Montreal Cognitive Assessment (range, 0 to 30, with higher scores indicating better cognitive ability) and the modified Rankin scale (range, 0 to 6, with higher scores indicating greater disability) at 3 months, and the CPC at 3 months.
在一项2×2析因设计的双盲、随机试验中,我们在院外心脏停搏(推测为心脏原因)后复苏成功的昏迷成人中比较了63 mm Hg和77 mm Hg两个平均动脉压目标;患者还被分配到两个氧合目标之一(在另外一篇论文中报告结果)。主要结局是由90日内的全因死亡或出院时脑功能分类(Cerebral Performance Category,CPC)3或4(范围,0~5,分类较高表明失能较严重;3或4表明严重失能或昏迷)构成的复合结局。次要结局包括48小时的神经元特异性烯醇化酶水平、90日内的全因死亡、3个月时的蒙特利尔认知评估评分(Montreal Cognitive Assessment,范围,0~30,评分较高表明认知能力较好)和改良Rankin量表评分(范围,0~6分,评分较高表明失能较严重)以及3个月时的CPC。
RESULTS
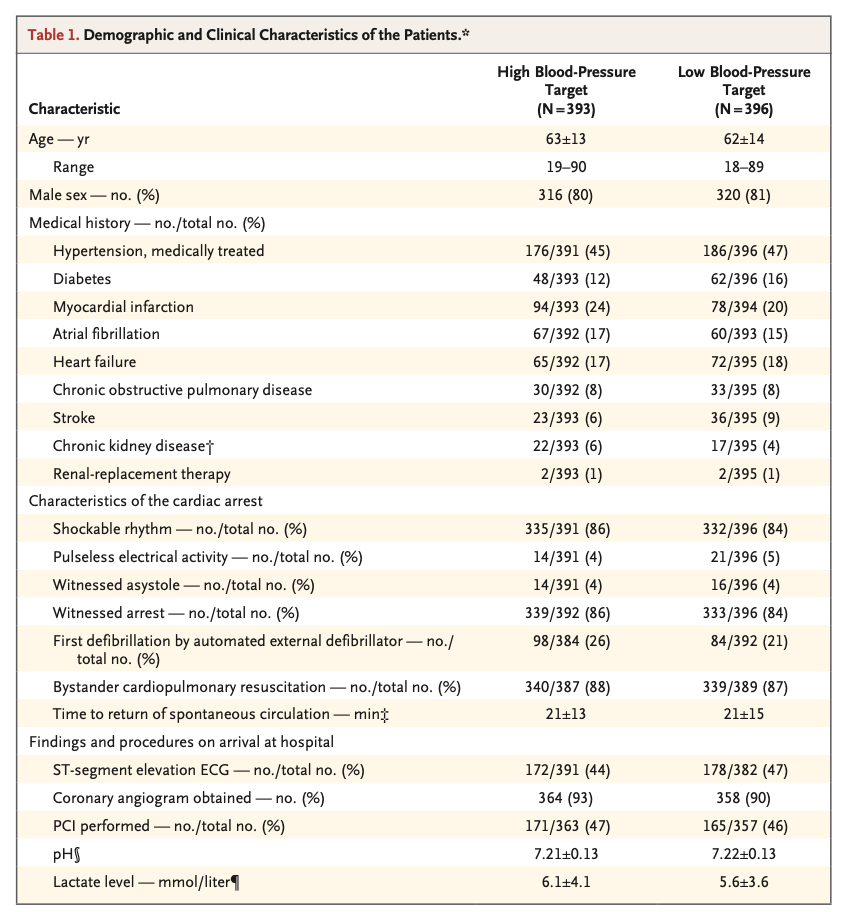
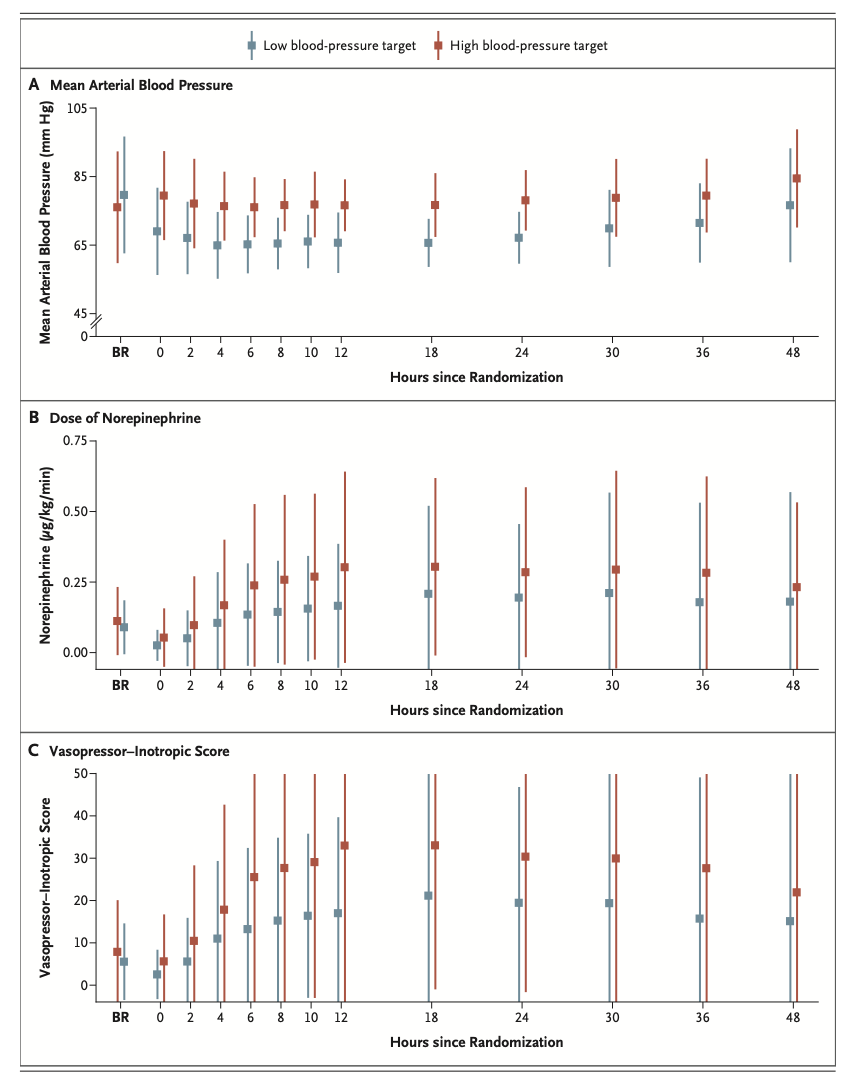
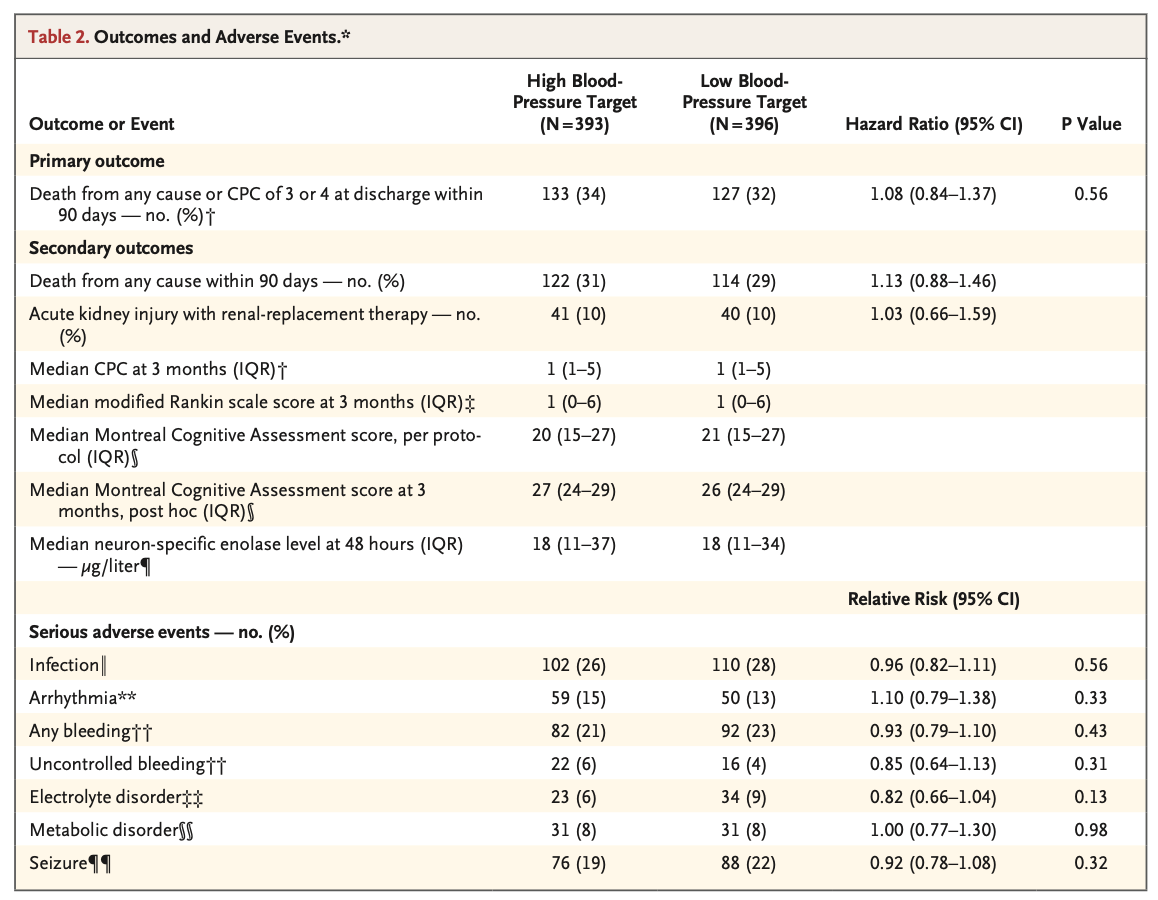
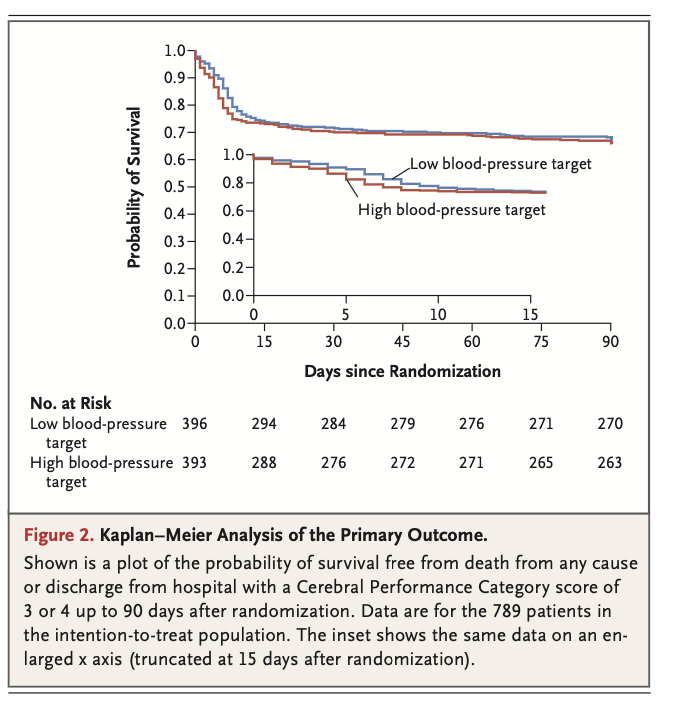
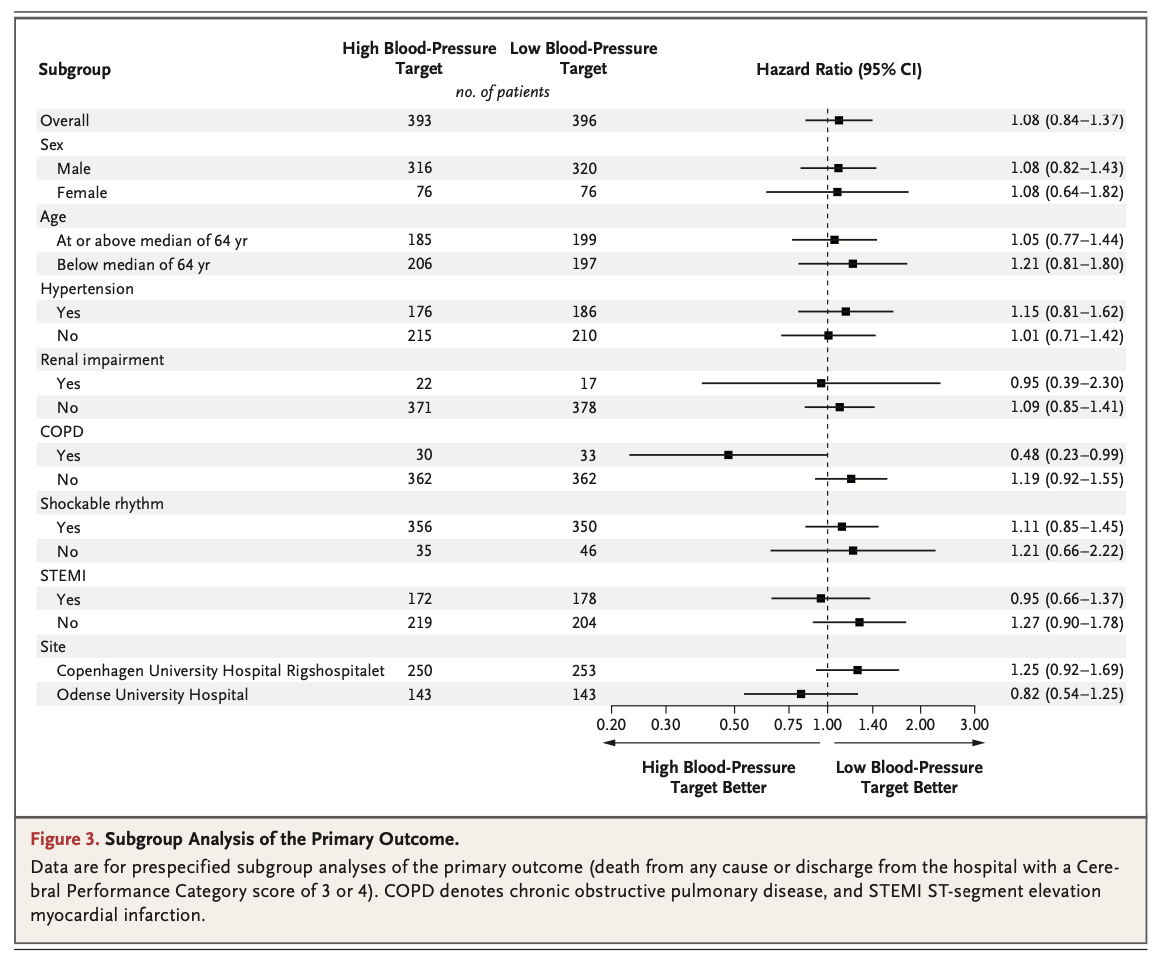
A total of 789 patients were included in the analysis (393 in the high-target group and 396 in the low-target group). A primary-outcome event occurred in 133 patients (34%) in the high-target group and in 127 patients (32%) in the low-target group (hazard ratio, 1.08; 95% confidence interval [CI], 0.84 to 1.37; P=0.56). At 90 days, 122 patients (31%) in the high-target group and 114 patients (29%) in the low- target group had died (hazard ratio, 1.13; 95% CI, 0.88 to 1.46). The median CPC was 1 (interquartile range, 1 to 5) in both the high-target group and the low-target group; the corresponding median modified Rankin scale scores were 1 (interquartile range, 0 to 6) and 1 (interquartile range, 0 to 6), and the corresponding median Montreal Cognitive Assessment scores were 27 (interquartile range, 24 to 29) and 26 (interquartile range, 24 to 29). The median neuron-specific enolase level at 48 hours was also similar in the two groups. The percentages of patients with adverse events did not differ significantly between the groups.
共计789例患者被纳入分析(高目标组393例,低目标组396例)。高目标组133例患者(34%)和低目标组127例患者(32%)发生了主要结局事件(风险比,1.08;95%置信区间[CI],0.84~1.37;P=0.56)。90日时,高目标组122例患者(31%)和低目标组114例患者(29%)已死亡(风险比,1.13;95% CI,0.88~1.46)。高目标组和低目标组的中位CPC均为1(四分位距,1~5);相应中位改良Rankin量表评分为1分(四分位距,0~6)和1分(四分位距,0~6),相应中位蒙特利尔认知评估评分为27分(四分位距,24~29)和26分(四分位距,24~29)。两组的48小时中位神经元特异性烯醇化酶水平也相似。两组间的不良事件发生率无显著差异。
CONCLUSIONS
Targeting a mean arterial blood pressure of 77 mm Hg or 63 mm Hg in patients who had been resuscitated from cardiac arrest did not result in significantly different percentages of patients dying or having severe disability or coma. (Funded by the Novo Nordisk Foundation; BOX ClinicalTrials.gov number, NCT03141099.)
对于心脏停搏后复苏成功的患者,将平均动脉压设定为77 mm Hg或63 mm Hg未导致死亡或者严重失能或昏迷的患者百分比出现显著差异。(由诺和诺德基金会[Novo Nordisk Foundation]资助;BOX在ClinicalTrials.gov注册号为NCT03141099。)
原创文章(本站视频密码:66668888),作者:xujunzju,如若转载,请注明出处:https://zyicu.cn/?p=13998

 微信扫一扫
微信扫一扫  支付宝扫一扫
支付宝扫一扫