Abstract
BACKGROUND
Perioperative bleeding is common in patients undergoing noncardiac surgery. Tranexamic acid is an antifibrinolytic drug that may safely decrease such bleeding.
围手术期出血常见于非心脏手术患者。氨甲环酸是一种抗纤溶药物,可安全减少此类出血。
METHODS
We conducted a trial involving patients undergoing noncardiac surgery. Patients were randomly assigned to receive tranexamic acid (1-g intravenous bolus) or placebo at the start and end of surgery (reported here) and, with the use of a partial factorial design, a hypotension-avoidance or hypertension-avoidance strategy (not reported here). The primary efficacy outcome was life-threatening bleeding, major bleeding, or bleeding into a critical organ (composite bleeding outcome) at 30 days. The primary safety outcome was myocardial injury after noncardiac surgery, nonhemorrhagic stroke, peripheral arterial thrombosis, or symptomatic proximal venous thromboembolism (composite cardiovascular outcome) at 30 days. To establish the noninferiority of tranexamic acid to placebo for the composite cardiovascular outcome, the upper boundary of the one-sided 97.5% confidence interval for the hazard ratio had to be below 1.125, and the one-sided P value had to be less than 0.025.
我们进行了一项涉及接受非心脏手术患者的试验。患者被随机分配在手术开始和结束时接受氨甲环酸(1g静脉推注)或安慰剂(此处报告),并使用部分析因设计,避免低血压或避免高血压策略(此处未报告)。主要疗效结局为第30天时危及生命的出血、大出血或关键器官出血(复合出血结局)。主要安全性结局为30天时非心脏手术后的心肌损伤、非出血性卒中、外周动脉血栓形成或症状性近端静脉血栓栓塞(复合心血管结局)。为了确定氨甲环酸在复合心血管结局方面非劣效于安慰剂,风险比的单侧97.5%置信区间上限必须低于1.125,单侧 P 值必须小于0.025。
RESULTS
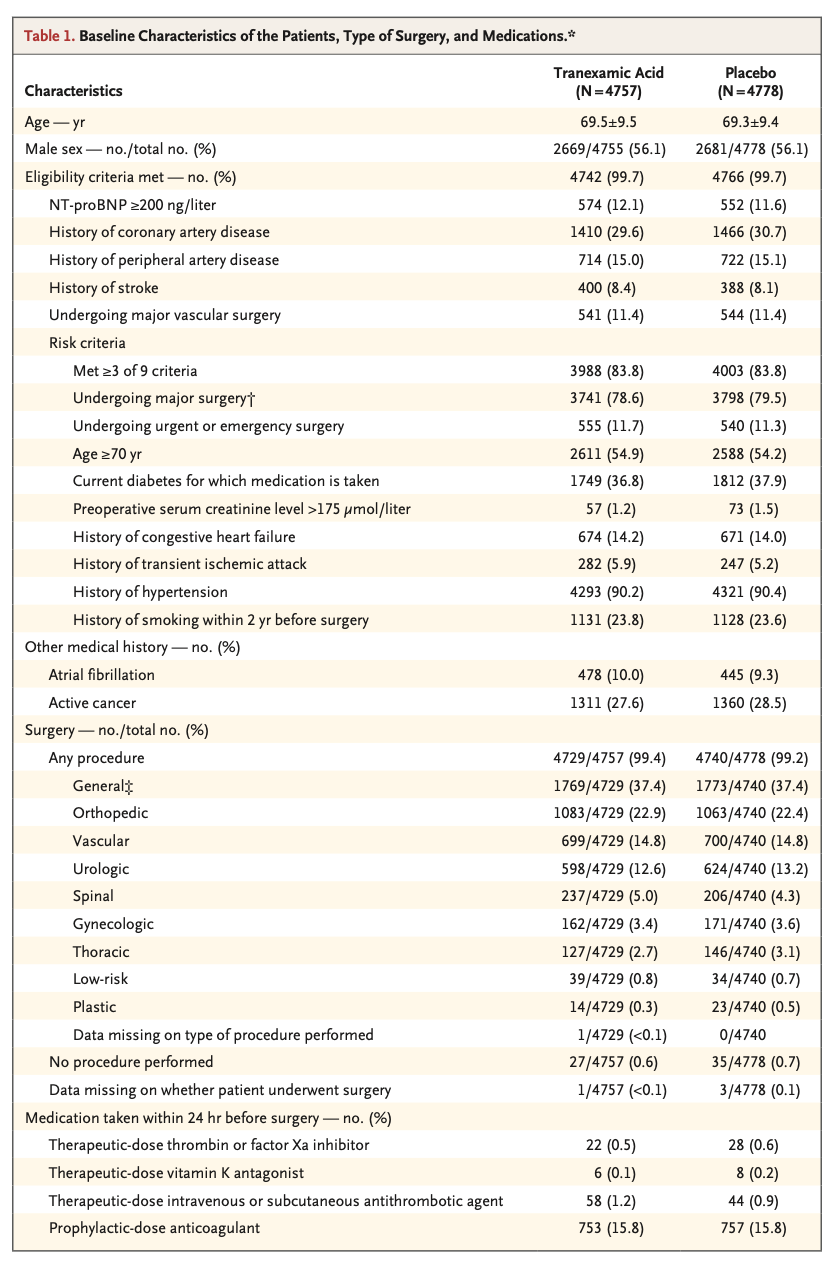
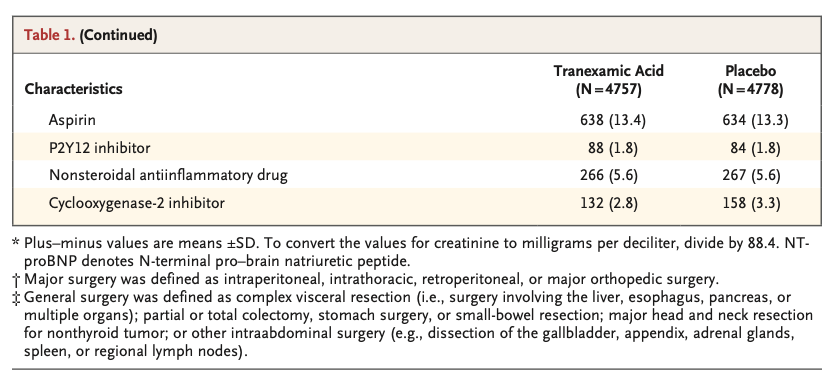
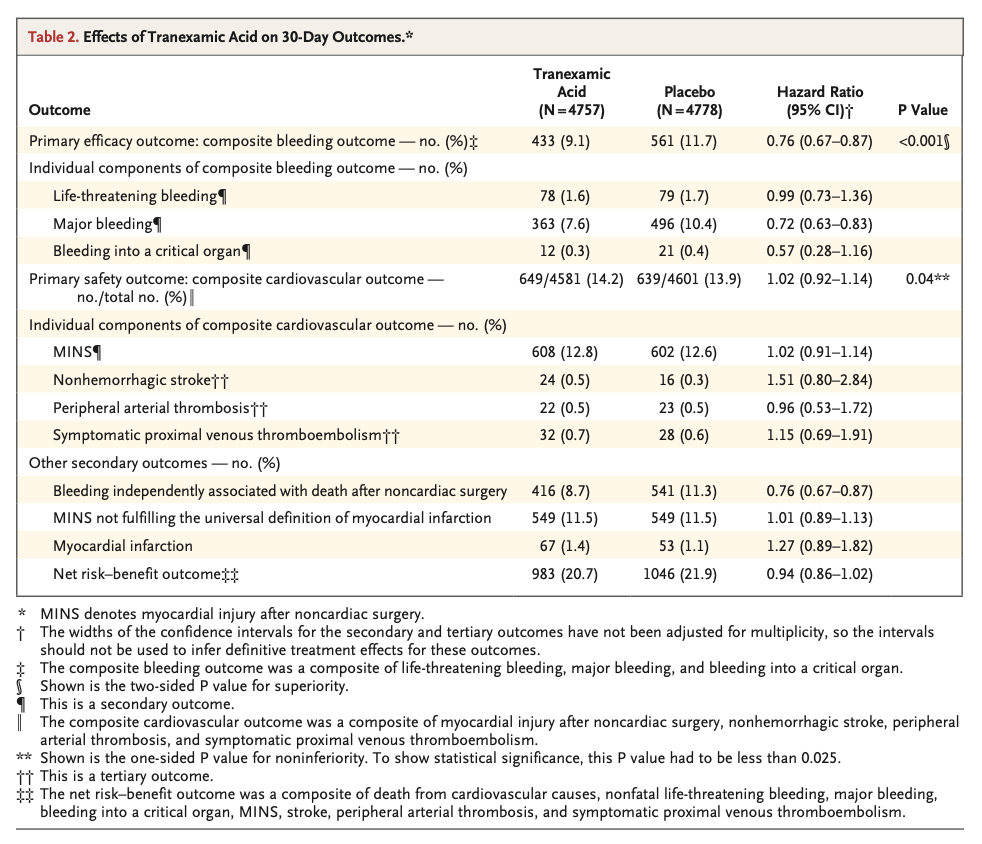
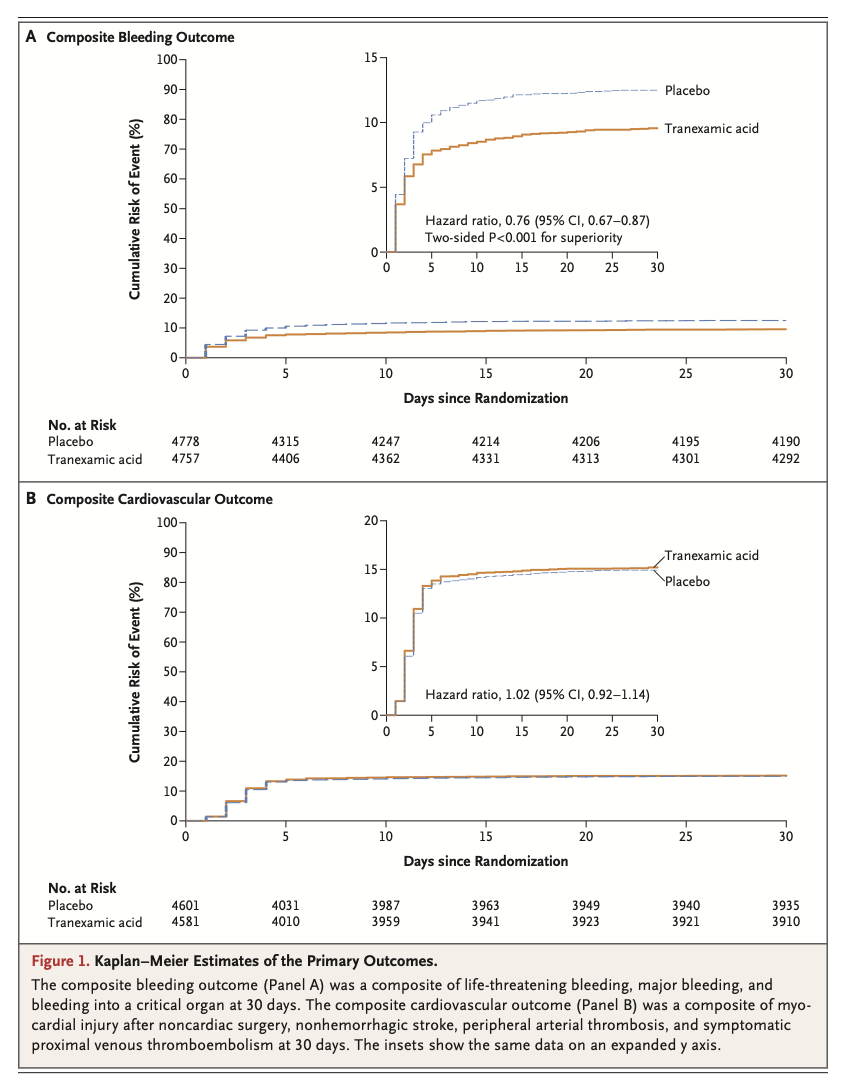
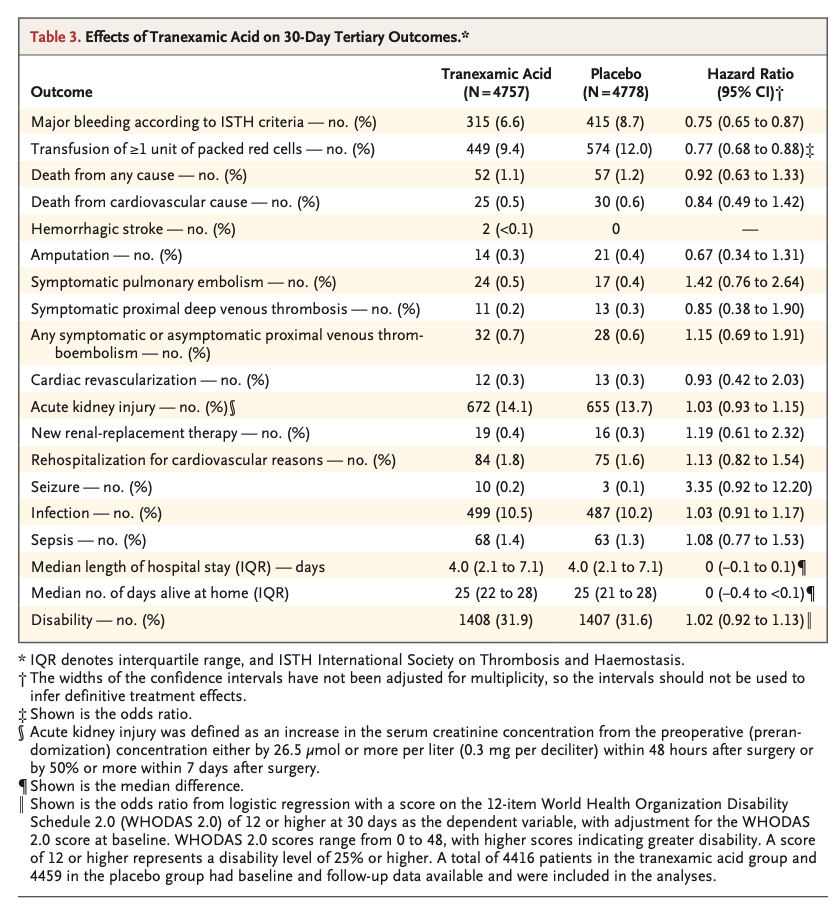
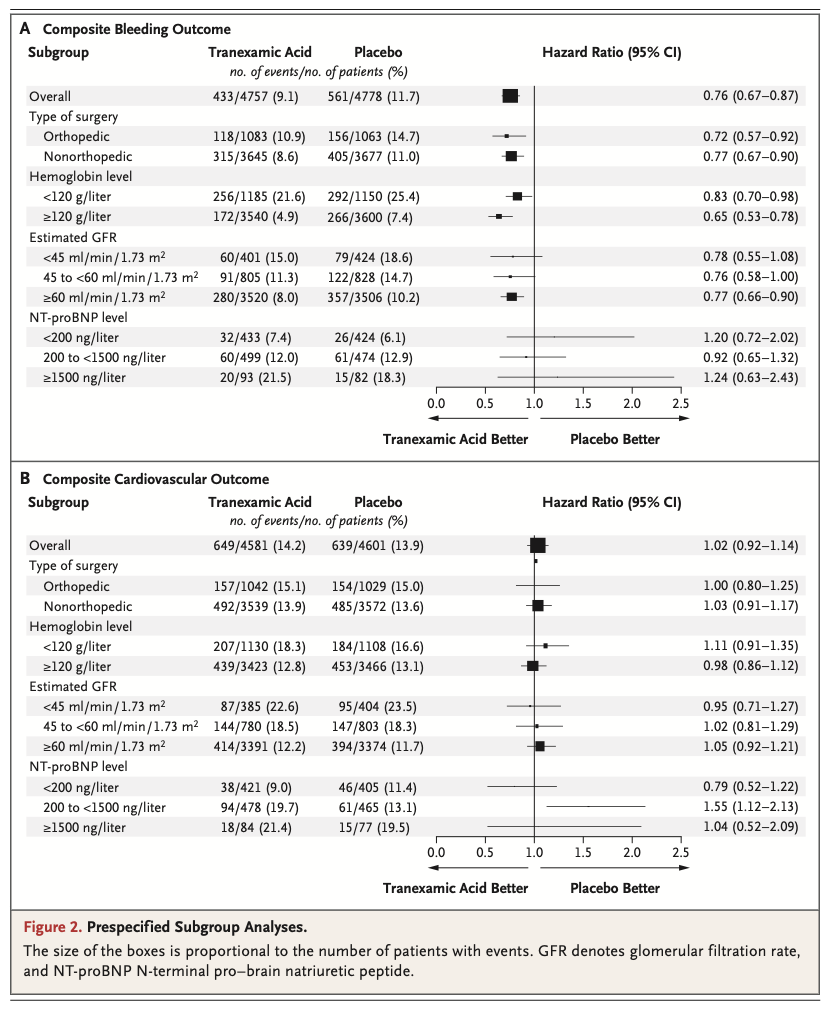
A total of 9535 patients underwent randomization. A composite bleeding outcome event occurred in 433 of 4757 patients (9.1%) in the tranexamic acid group and in 561 of 4778 patients (11.7%) in the placebo group (hazard ratio, 0.76; 95% confidence interval [CI], 0.67 to 0.87; absolute difference, −2.6 percentage points; 95% CI, −3.8 to −1.4; two-sided P<0.001 for superiority). A composite cardiovascular outcome event occurred in 649 of 4581 patients (14.2%) in the tranexamic acid group and in 639 of 4601 patients (13.9%) in the placebo group (hazard ratio, 1.02; 95% CI, 0.92 to 1.14; upper boundary of the one-sided 97.5% CI, 1.14; absolute difference, 0.3 percentage points; 95% CI, −1.1 to 1.7; one-sided P=0.04 for noninferiority).
共有9535例患者接受了随机化。氨甲环酸组4757例患者中的433例 (9.1%) 和安慰剂组4778例患者中的561例 (11.7%) 发生复合出血结局事件(风险比,0.76;95%置信区间 [CI],0.67至0.87;绝对差异,-2.6个百分点;95%CI,-3.8至-1.4;优效性双侧P < 0.001)。氨甲环酸组4581例患者中的649例 (14.2%) 和安慰剂组4601例患者中的639例 (13.9%) 发生复合心血管结局事件(风险比,1.02;95%CI,0.92-1.14;单侧97.5%CI上限,1.14;绝对差异,0.3个百分点;95%CI,-1.1至1.7;非劣效性单侧P = 0.04)。
CONCLUSIONS
Among patients undergoing noncardiac surgery, the incidence of the composite bleeding outcome was significantly lower with tranexamic acid than with placebo. Although the between-group difference in the composite cardiovascular outcome was small, the noninferiority of tranexamic acid was not established. (Funded by the Canadian Institutes of Health Research and others; POISE-3 ClinicalTrials.gov number, NCT03505723. opens in new tab.)
在接受非心脏手术的患者中,氨甲环酸组复合出血结局的发生率显著低于安慰剂组。尽管复合心血管结局的组间差异较小,但尚未确定氨甲环酸的非劣效性。
原创文章(本站视频密码:66668888),作者:xujunzju,如若转载,请注明出处:https://zyicu.cn/?p=13116

 微信扫一扫
微信扫一扫  支付宝扫一扫
支付宝扫一扫