
Cryptococcosis is a fungal disease caused by Cryptococcus neoformans and Cryptococcus gattii. By inhalation and subsequent pulmonary infection, it may disseminate to the CNS and cause meningitis or meningoencephalitis. Most cases occur in immunosuppressed hosts, including patients with HIV/AIDS, patients receiving immunosuppressing drugs, and solid organ transplant recipients. However, cryptococcosis also occurs in individuals with apparently healthy immune systems. A growing number of cases are caused by C gattii, with infections occurring in both immunosuppressed and immunocompetent individuals. In the majority of documented cases, treatment of C gattii infection of the CNS requires aggressive management of raised intracranial pressure along with standard antifungal therapy. Early cerebrospinal fluid evacuation is often needed through placement of a percutaneous lumbar drain or ventriculostomy. Furthermore, pharmacological immunosuppression with a high dose of dexamethasone is sometimes needed to ameliorate a persistently increased inflammatory response and to reduce intracranial pressure. In this Grand Round, we present the case of an otherwise healthy adolescent female patient, who, despite aggressive management, succumbed to C gattii meningoencephalitis. We also present a review of the existing literature and discuss optimum clinical management of meningoencephalitis caused by C gattii.
隐球菌病是由新型隐球菌和格特隐球菌引起的真菌病。通过吸入和随后的肺部感染,可播散至CNS,引起脑膜炎或脑膜脑炎。大多数病例发生在免疫抑制宿主,包括HIV/AIDS患者、接受免疫抑制药物的患者和实体器官移植受者。然而,隐球菌病也发生在免疫系统表面健康的个体中。越来越多的病例是由格特隐球菌引起的,免疫抑制和免疫功能正常的个体都会发生感染。在大多数记录的病例中,CNS格特隐球菌感染的治疗需要积极管理颅内压升高以及标准抗真菌治疗。通常需要通过放置经皮腰椎引流管或脑室引流管进行早期脑脊液排空。此外,有时需要使用高剂量地塞米松进行药物免疫抑制,以改善持续增加的炎症反应并降低颅内压。在本轮病例中,我们报告了1例其他方面健康的青少年女性患者,尽管进行了积极治疗,但仍死于格特隐球菌脑膜脑炎。我们还对现有文献进行了综述,并讨论了格特隐球菌引起脑膜脑炎的最佳临床管理。
Introduction
Cryptococcosis is one of the most common invasive fungal diseases in human beings, with more than 1 million cases per year and around 650000 deaths in sub-Saharan Africa. Of the more than 30 species of the genus Cryptococcus, Cryptococcus neoformans and Cryptococcus gattii are the only species that are commonly pathogenic because of their ability to grow at 37°C and the presence of other virulence factors such as production of melanin and a protective capsule. The epidemiology and clinical features of infections caused by C neoformans have been previously described. Infections occur worldwide, affecting predominantly patients with HIV/AIDS or other immunocompromising conditions, although infections do occur among apparently immmunocompetent individuals.
隐球菌病是人类最常见的侵袭性真菌病之一,在撒哈拉以南非洲每年有超过100万病例,约有65万人死亡。在隐球菌属的30多种菌种中,新型隐球菌和格特隐球菌是唯一常见致病的菌种,因为它们能够在37 ℃下生长,并存在其他毒力因子如产生黑色素和保护性荚膜。以前曾描述过新生隐球菌引起的感染的流行病学和临床特征。感染发生在世界范围内,主要影响HIV/AIDS或其他免疫功能低下疾病患者,尽管感染确实发生在明显免疫功能正常的个体中。
Many aspects of the epidemiology and clinical features of infections caused by C gattii are relatively less well defined. C gattii is a fungal pathogen that grows preferentially in soil around various kinds of trees. Similar to C neoformans, it causes pulmonary and CNS disease in people. The initial recognition of C gattii as a pathogen was reported in a patient with a lumbar tumour by pathologist Ferdinand Curtis in 1896. Historically, most cases due to C gattii have been seen in tropical and subtropical regions, but it is now regarded as an emerging fungal pathogen in other geographical settings.9 Cases of C gattii meningoencephalitis occurring among otherwise healthy, immunocompetent individuals have predominated in the literature. However, recent reports from the USA have shown that those infected frequently have some underlying condition that could potentially be associated with immunosuppression. C gattii tend to produce severe CNS manifestations, including meningitis, encephalitis, or, more frequently, meningoencephalitis. These manifestations might lead to excessive neurological morbidity due to the associated intracranial hypertension.
格特隐球菌引起的感染的流行病学和临床特征的许多方面相对不太明确。格特隐球菌是一种真菌病原体,优先生长在各种树木周围的土壤中。与新型隐球菌相似,它会引起人的肺部和CNS疾病。1896年病理学家Ferdinand Curtis报道了1例腰椎肿瘤患者最初认识到格特隐球菌是一种病原体。历史上,大多数格特隐球菌所致病例见于热带和亚热带地区,但目前在其他地理环境中被视为一种新出现的真菌病原体。文献报道9例发生于其他方面健康、免疫功能正常个体中的格特隐球菌脑膜脑炎占主导地位。然而,最近来自美国的报告显示,那些经常感染的人有一些可能与免疫抑制相关的基础疾病。格特隐球菌倾向于产生严重的CNS表现,包括脑膜炎、脑炎或更常见的脑膜脑炎。由于相关的颅内高压,这些表现可能导致过度的神经系统疾病。
C gattii was previously thought to be a subtype of C neoformans (subtype B and C, referring to capsular antigens), but is now recognised as a unique species. The species is divided into four unique molecular types (variety gattii; VGI-IV). There is endemicity of VGI and VGII strains in Australia, VGII and VGIII strains in South America, VGI strains in India, and VGIV strains in Africa. In the USA, cases of C gattii have been noted in southern California and Hawaii. Typing of isolates recovered from human beings and animals in those regions suggested similarity to other strains arising in more tropical regions, specifically VGI and VGIII. Since 2004, an outbreak of infection has been identified in the Pacific northwest region in North America, involving primarily clonal VGII strains (clonal VGIIa and VGIIb in Canada, and clonal VGIIc in the USA). These isolates, first recognised on Vancouver Island, have now been documented to have expanded onto western mainland Canada as well as several Pacific northwestern states in the USA. Many of these cases have presented predominantly with respiratory symptoms and have occurred in immunocompromised hosts (38% of British Columbia cases and 59% of the USA cases; table). Moreover, since 2009, more than 25 autochthonous (non-outbreak) cases of C gattii have been documented in other parts of the USA, the most common molecular types being VGI or VGIII.
格特隐球菌以前被认为是新生隐球菌的一个亚型(B和C亚型,指荚膜抗原),但现在被认为是一个独特的物种。该物种分为4种独特的分子类型(品种格特;VGI-IV)。澳大利亚存在VGI和VGII株流行,南美洲存在VGII和VGIII株流行,印度存在VGI株流行,非洲存在VGIV株流行。在美国,在南加州和夏威夷观察到格特隐球菌病例。从这些地区的人和动物中回收的分离株的分型表明与更热带地区的其他菌株相似,尤其是VGI和VGIII。自2004年以来,在北美太平洋西北地区发现了感染暴发,主要涉及克隆VGII菌株(加拿大的克隆VGIIa和VGIIb,美国的克隆VGIIc)。这些分离株首次在温哥华岛被认识,目前已被证实已扩展到加拿大大陆西部以及美国几个太平洋西北部州。其中许多病例主要表现为呼吸道症状,并发生在免疫功能低下的宿主中(38%的不列颠哥伦比亚省病例和59%的美国病例;表)。此外,自2009年以来,在美国其他地区记录了超过25例格特隐球菌本土(非暴发)病例,最常见的分子类型为VGI或VGIII。
Much of our knowledge on cryptococcosis has been derived from studies focused on C neoformans infection in people with HIV. We now appreciate several unique features of CNS disease caused by C gattii. Herein, we present an illustrative case and review the existing medical literature to address the optimum medical management of meningoencephalitis caused by C gattii.
我们对隐球菌病的大部分知识来源于对HIV感染者新生隐球菌感染的研究。我们现在了解由格特隐球菌引起的CNS疾病的几个独特特征。在此,我们介绍了一例说明性病例,并对现有的医学文献进行了综述,以解决格特隐球菌引起的脑膜脑炎的最佳医学管理。
Case description
A previously healthy 18-year-old woman was admitted to a hospital in Georgia, USA, with a 1-week history of severe headaches, altered mental status, and new onset seizures. She was a college student with no previous medical history, including no history of recurrent infections, no travel history outside the state, and was not taking any medications before her admission to the hospital. Her parents and sister were healthy. On admission, CT scan imaging of the head without contrast was unremarkable, and because of intermittent fever episodes and headache during a 5-day inpatient course, she underwent a lumbar puncture. The finding of budding yeast in the cerebrospinal fluid (CSF) as well as CSF pleocytosis prompted transfer of the patient to our regional hospital (Phoebe Putney Memorial Hospital, Albany, GA, USA) for management of fungal meningitis. On admission to the hospital, the patient suffered a generalised tonic- clonic seizure. She was stuporous and unable to provide any medical history. Her blood pressure was 130/77 mm Hg, pulse 106 beats per min, respiratory rate 18 breaths per min, and her temperature was 37°C. Her neck was supple with no signs of meningeal irritation. She did not have any skin lesions or rashes and was able to open her eyes to painful stimuli. Her pupils were symmetrical and equally reactive with intact oculocephalic and corneal reflexes. No facial asymmetry or grimace was noted. She had normal tone in her upper and lower limbs and withdrawal to painful stimuli. Her lung, cardiac, and abdominal examination were judged normal.
患者女,18岁,既往体健,因剧烈头痛、精神状态改变、新发癫痫发作1周入住美国乔治亚州某医院。她是一名大学生,既往无病史,包括无反复感染史,无州外旅行史,入院前未服用任何药物。她的父母和姐姐都很健康。入院时,无造影剂的头部CT扫描成像无明显异常,由于5天住院期间间歇性发热发作和头痛,她接受了腰椎穿刺。脑脊液(CSF)中发现芽殖酵母以及CSF细胞增多促使患者转至我们的地区医院(Phoebe Putney Memorial hospital,Albany,GA,USA)治疗真菌性脑膜炎。入院时,患者发生全身强直阵挛性发作。患者昏迷,无法提供任何病史。血压130/77 mmHg,脉搏106次/min,呼吸18次/min,体温37 ℃。颈部柔软,无脑膜刺激征。她没有任何皮肤病变或皮疹,能够对疼痛刺激睁开眼睛。她的瞳孔对称,反应相同,眼头反射和角膜反射完整。未观察到面部不对称或鬼脸。她的上肢和下肢张力正常,对疼痛刺激退缩。她的肺、心脏和腹部检查被判定为正常。
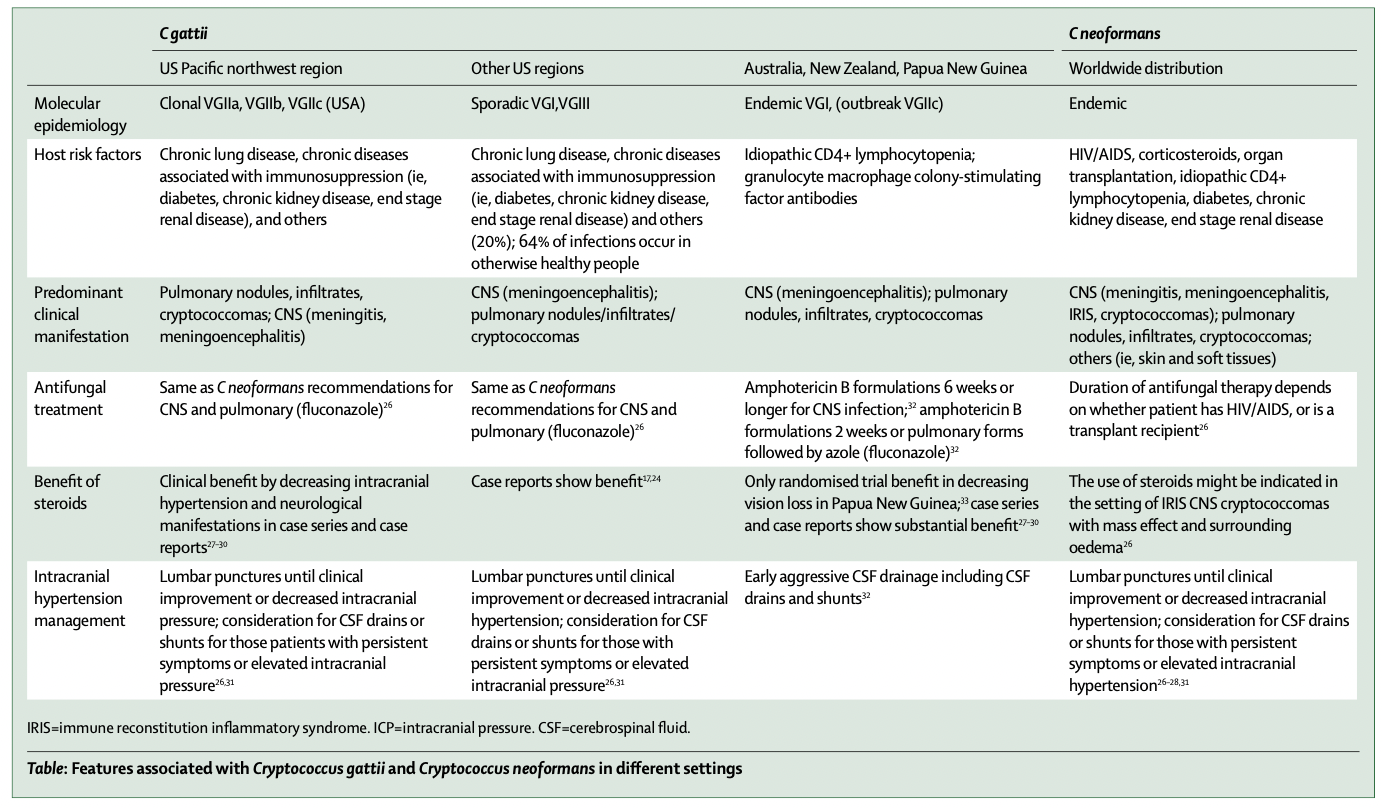
The patient was admitted to the intensive care unit, and because of progressive neurological deterioration she needed endotracheal intubation with ventilator support. A lumbar puncture was repeated, disclosing an opening pressure exceeding 60 cm of water. A lumbar drain was placed for CSF drainage and pressure monitoring. CSF analysis revealed a glucose concentration of 3·22 mmol/L, protein concentrations of 2·43 g/L and 860 white blood cells per field (61% mononuclear cells). The titre of cryptococcal antigen by enzyme immunoassay of CSF was greater than 1/512. Cryptococcus spp was isolated in CSF standard fungal culture media, while blood and urine cultures revealed no growth. An ELISA for HIV was negative, and her chest radiograph did not show evidence of any abnormalities. Liposomal amphotericin B (5 mg/kg) and flucytosine were given immediately. An MRI of the brain revealed multiple non-enhancing cystic masses within the bilateral basal ganglia and head of the caudate lobes, consistent with gelatinous pseudocysts (dilated Virchow-Robin perivascular spaces; figure 1).
患者入住重症监护室,由于进行性神经功能恶化,她需要气管插管和呼吸机支持。重复腰椎穿刺,显示开放压力超过60 cmH20。放置腰椎引流管进行CSF引流和压力监测。CSF分析显示葡萄糖浓度为3.22 mmol/L,蛋白浓度为2.43 g/L,每个视野860个白细胞(61%单核细胞)。CSF酶免疫法隐球菌抗原滴度大于1/512。在CSF标准真菌培养基中分离出隐球菌属,而血液和尿液培养显示无生长。HIV ELISA为阴性,其胸片未显示任何异常证据。立即给予两性霉素B脂质体(5 mg/kg)和氟胞嘧啶。脑部MRI显示双侧基底神经节和尾状叶头部内多发性无增强囊性肿块,与胶状假性囊肿一致(Virchow-Robin血管周围间隙扩张;图1)。
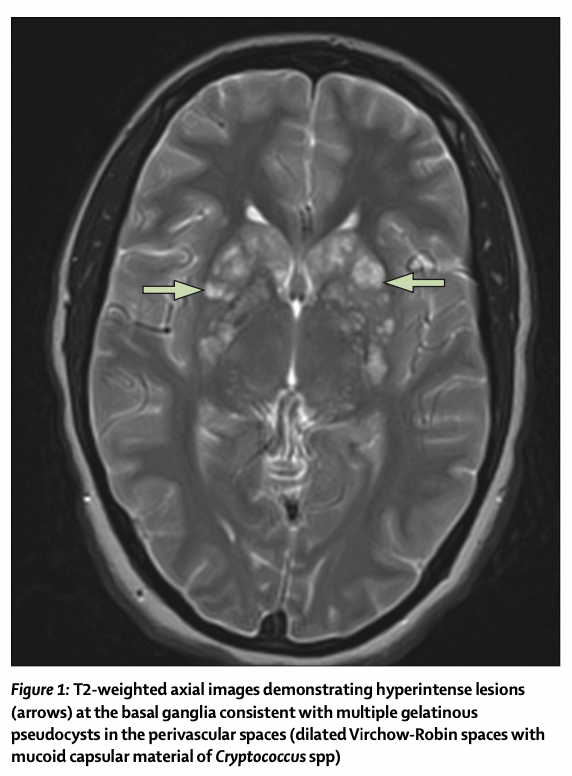
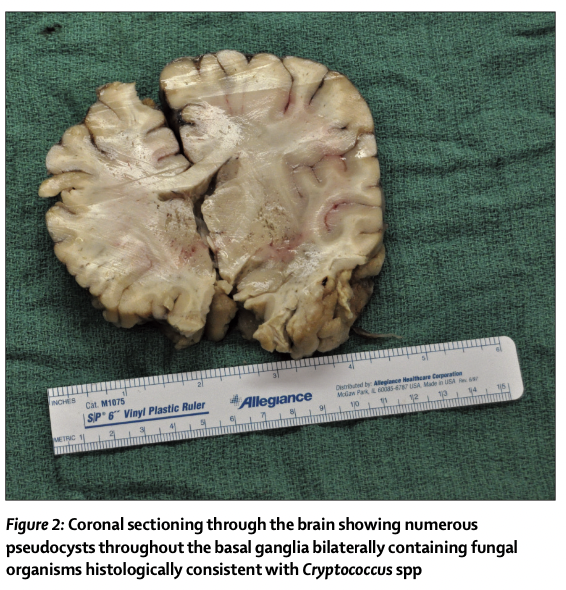
Despite aggressive intensive care support, the patient died about 48 hours after admission. A necropsy revealed marked asymmetry of the cerebral hemi- spheres, with midline shift from left to right and thickened meninges. Downward displacement of brainstem and cerebellum also showed evidence of central and tonsillar herniation (figure 2). Microscopic examination revealed many pseudocysts bilaterally throughout the basal ganglia, which contained multiple fungal organisms histologically consistent with Cryptococcus spp (figure 3). Focal areas of inflammation, oedema, and haemorrhage were found in areas where these organisms appear to be diffusely infiltrating the brain parenchyma (thalamus, periventricular white matter, midbrain, and the cerebellum). These areas were defined as small, and in some areas, confluent cryptococcomas (cryptococci inside parenchyma with associated oedema and inflammation).39,40 The actual number and distribution of lesions that were identified pathologically were underestimated by MRI images.
尽管进行了积极的重症监护支持,但患者在入院后约48小时死亡。尸检显示大脑半球明显不对称,中线从左移至右移,脑膜增厚。脑干和小脑向下移位也显示出中央和扁桃体疝的证据(图2)。显微镜检查显示整个基底神经节双侧有许多假性囊肿,其中含有多种组织学上与隐球菌属一致的真菌微生物(图3)。在这些微生物似乎弥漫性浸润脑实质的区域(丘脑、脑室周围白质、中脑和小脑)发现炎症、水肿和出血的局灶区域。这些区域被定义为小的,在某些区域,融合性隐球菌(实质内的隐球菌伴相关水肿和炎症)。MRI图像低估了病理确定的病变的实际数量和分布。
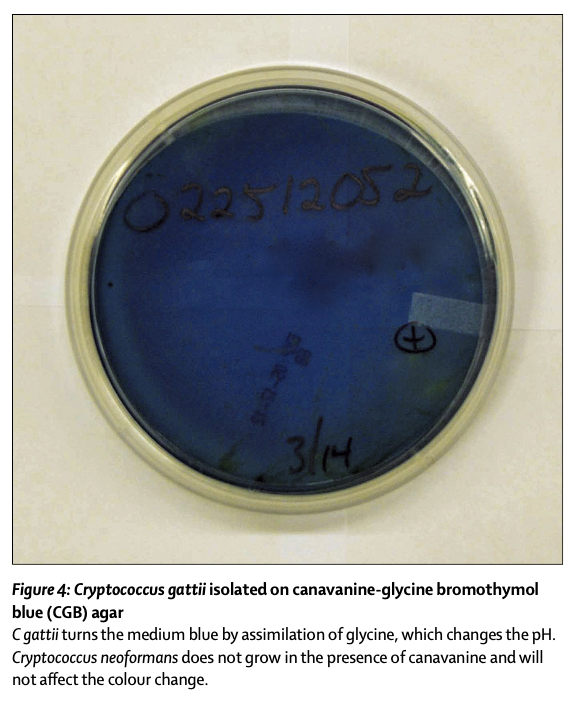
Our initial management focused on treating her case as C neoformans presenting in a patient without HIV, with the use of amphotericin B and flucytosine, in accordance with treatment guidelines. We concomitantly addressed her raised intracranial pressure by placing a lumbar drain to drain CSF and monitoring her intracranial pressure. When she began to deteriorate despite these interventions, she received a dose of 4 mg of dexamethasone. At this point, on the basis of her progressive course and MRI findings, we also suspected the possibility of C gattii meningoencephalitis.24 Before her death, vascular flow studies with 99mTc pertechnetate were consistent with photopenia, which, in turn, was consistent with decreased vascular supply to the cerebral hemispheres and diencephalon. 3 days later, culture of her CSF on agar containing canavianine, glycine, and bromothymol blue (CGB) (figure 4) and dihydroxy- phenylalanine/birdseed agar revealed growth of C gattii.41 Multilocus sequence typing conducted at the Fungal Reference Laboratory at the US Centers for Disease Control and Prevention identified C gattii molecular type VGIII, with the isolate having genetic similarity to other isolates from patients residing in the southeastern USA.
我们最初的治疗重点是根据治疗指南,使用两性霉素B和氟胞嘧啶将她的病例治疗为无HIV患者的新型隐球菌。我们通过放置腰椎引流管引流CSF并监测她的颅内压,同时解决了她的颅内压升高。尽管接受了这些干预措施,但病情仍开始恶化时,患者接受了4 mg地塞米松治疗。此时,根据她的进展过程和MRI表现,我们也怀疑格特隐球菌脑膜脑炎的可能性。在她死亡前,99mTc高锝酸盐的血管血流研究符合光减少,进而符合大脑半球和间脑的血管供应减少。3天后,在含有canavianine、甘氨酸、溴百里酚蓝(CGB)(图4)和二羟基苯丙氨酸/鸟种子琼脂显示格特隐球菌生长。在美国疾病控制和预防中心的真菌参考实验室进行的多位点序列分型鉴定出格特C分子VGIII型,分离株与居住在美国东南部患者的其他分离株具有遗传相似性。
Review and discussion
Cryptococcal meningoencephalitis is the most severe clinical manifestation caused by C gattii. We postulate that infection of the CNS by C gattii molecular type VGIII in our patient produced a severe inflammatory response. This was demonstrated by the histopathological evidence of severe meningoencephalitis, with substantial thickening of her meninges, diffuse infiltration of Cryptococci spp in the parenchyma, and associated inflammation and oedema, similar to necropsy reports of individuals without HIV. Elevated titres of cryptococcal antigen in the CSF, positive CSF culture, and high CSF pleocytosis were markers of high fungal burden and severe inflammation.
隐球菌性脑膜脑炎是由格特隐球菌引起的最严重的临床表现。我们假定在我们的患者中,格特C分子类型VGIII感染CNS产生了严重的炎症反应。与无HIV个体的尸检报告相似,重度脑膜脑炎的组织病理学证据证实了这一点,脑膜显著增厚,实质中隐球菌属弥漫性浸润,并伴有炎症和水肿。CSF中隐球菌抗原滴度升高、CSF培养阳性和高CSF细胞增多是高真菌负荷和重度炎症的标志物。
We postulate that the elevated intracranial pressure experienced by our patient jointly resulted from an increased volume of brain tissue, caused by parenchymal inflammation and interstitial oedema (identified in necropsy), and an augmented CSF volume, caused by outflow resistance. Decreased outflow of CSF was possibly triggered by the presence of yeast and its capsular polysaccharide at the arachnoid granulations, where arachnoid villus cells resorb CSF and meningeal inflammation. Compensatory mechanisms that ensued to accommodate for the increased brain CSF volume were exhausted, leading to mass effect producing pathological consequences by displacing surrounding tissue and cerebral herniation. Although there are important pathogenic differences between acute bacterial meningitis and cryptococcal meningoencephalitis, we can infer from evidence of the association of acute bacterial meningitis and lumbar puncture that most reports of herniation occur shortly after a lumbar puncture. Thus, our patient’s progressive neurological decline and the timing of lumbar puncture relative to her time of death, suggest that iatrogenic cerebral herniation syndrome due to CSF drainage was unlikely. Only a scant number of necropsy reports are available to allow us to assess the rate of occurrence of brain herniation syndromes associated with CNS cryptococcosis.
我们假定,我们的患者经历的颅内压升高共同是由脑实质炎症和间质水肿(在尸检中确定)引起的脑组织体积增加和由流出阻力引起的CSF体积增加所致。CSF流出减少可能是由蛛网膜颗粒处存在酵母及其荚膜多糖触发的,蛛网膜绒毛细胞可吸收CSF和脑膜炎症。随后适应脑CSF体积增加的代偿机制耗尽,导致占位效应,通过置换周围组织和脑疝产生病理后果。尽管急性细菌性脑膜炎和隐球菌性脑膜脑炎之间存在重要的致病差异,但我们可以从急性细菌性脑膜炎和腰椎穿刺相关性的证据中推断,大多数疝形成报告发生在腰椎穿刺后不久。因此,我们的患者的进行性神经功能下降和相对于其死亡时间的腰椎穿刺时间,表明CSF引流不太可能导致医源性脑疝综合征。只有很少数量的尸检报告允许我们评估与CNS隐球菌病相关的脑疝综合征的发生率。
While ongoing research continues to elucidate the epidemiology, pathogenesis, and clinical expression of C gattii infection in human beings, evidence from animal models and clinical reports suggest that susceptibility to infection and clinical expression depends on host, site of infection, strain, and endemic or sporadic versus outbreak settings (table). C gattii can affect both immunocompromised as well as otherwise immuno- competent individuals and can affect both the lungs and the CNS. Host risk factors for infection include HIV/ AIDS, idiopathic CD4+ lymphopenia, chronic lung diseases, or immunosuppression associated with diabetes or chronic kidney disease. Many of the individuals previously identified as otherwise immuno- competent might actually have a predisposing phagocyte dysfunction, as suggested by the recent identification of antibodies against granulocyte macrophage colony- stimulating factor in patients with C gattii meningo- encephalitis.
虽然正在进行的研究继续阐明人类格特隐球菌感染的流行病学、发病机制和临床表现,但动物模型和临床报告的证据表明,感染易感性和临床表现取决于宿主、感染部位、菌株以及地方性或散发性与暴发环境(表)。格特隐球菌可影响免疫功能低下和其他免疫功能正常的个体,并可影响肺部和CNS。感染的宿主风险因素包括HIV/AIDS、特发性CD4 + 淋巴细胞减少症、慢性肺病或与糖尿病或慢性肾病相关的免疫抑制。正如最近在格特隐球菌脑膜脑炎患者中发现的抗粒细胞巨噬细胞集落刺激因子的抗体所提示的,许多先前被鉴定为具有其他免疫活性的个体实际上可能具有易感吞噬细胞功能障碍。
Evidence suggests that in some settings, and depending on the host factors and strain, C gattii tend to produce more severe CNS manifestations compared with C neoformans. Reports from Australia and Papua New Guinea first suggested that CNS disease caused by C gattii is characterised by severe CNS inflammation, high intracranial pressure complications, crypto- coccomas, and poor prognoses in terms of neurological sequelae. During the past few years, reports have also suggested that the immune response to C gattii among individuals without HIV and who are otherwise immunocompetent can paradoxically present with more severe CNS disease, with an immune reconstitution inflammatory syndrome (IRIS)-like syndrome, or with sterile arachnoiditis. The molecular type and strain are probably important in the spectrum of disease caused by C gattii. As illustrated by case series from Australia, Papua New Guinea, and Colombia, infection with C gattii molecular types VGI, VGII, and non-outbreak strain VGII usually presents as severe meningo- encephalitis. Similarly, most sporadic (non-outbreak) cases reported from the USA that are caused by either VGI or VGIII types frequently present with severe meningoencephalitis, similar to our patient. However, infections in the Pacific northwest region of the USA and British Columbia, Canada, which are caused mostly by VGIIa, manifest more likely as pulmonary disease.
有证据表明,在某些情况下,根据宿主因素和菌株,与新生隐球菌相比,格特隐球菌倾向于产生更严重的CNS表现。来自澳大利亚和巴布亚新几内亚的报告首先表明,格特隐球菌引起的CNS疾病的特征是严重的CNS炎症、高颅内压并发症、隐球菌和神经系统后遗症方面的不良预后。在过去的几年中,也有报道认为,在没有HIV和其他免疫功能正常的个体中,对格特隐球菌的免疫反应可以矛盾地表现为更严重的CNS疾病,伴有免疫重建炎症综合征(IRIS)样综合征,或伴有无菌性蛛网膜炎。分子类型和菌株可能在格特立克次体引起的疾病谱中很重要。如澳大利亚、巴布亚新几内亚和哥伦比亚的病例系列所示,格特C分子类型VGI、VGII和非暴发菌株VGII感染通常表现为重度脑膜脑炎。同样,美国报告的由VGI或VGIII型引起的大多数散发(非暴发)病例通常表现为重度脑膜脑炎,与我们的患者相似。然而,主要由VGIIa引起的美国太平洋西北地区和加拿大不列颠哥伦比亚省的感染更可能表现为肺部疾病。
Mechanism of CNS disease
Most of what is known about the pathogenesis of cryptococcosis has been derived from studies of C neoformans. Cryptococcus spp establish pulmonary infection when spores or dessicated cells are inhaled. Once in the lung, the yeast can reach the bloodstream and travel to the CNS, entering the CSF via transcellular migration across the microvascular endothelium of the blood–brain barrier. Other mechanisms for crossing of the blood–brain barrier include a so-called Trojan horse mechanism, whereby Cryptococcus spp cross the endothelium by hiding inside mononuclear cells or by using paracellular migration pathways. Three hypotheses have been proposed to explain fungal neurotropism. First, neuronal substrates in the basal ganglia are thought to promote cryptococcal growth and survival. The CNS, and particularly the perivascular spaces (Virchow-Robin spaces), might serve as a niche for Cryptococcus spp. Our patient showed evidence of Cryptococcus spp infection within perivascular spaces of the parenchyma supplied by the perforating lenticulostriate branches of the middle cerebral arteries. Further signs of infection were found at other perivascular spaces, including the thalamus, peri- ventricular white matter, cerebellum, and midbrain (figure 2). Second, there seem to be specific neuronal receptors that might attract this fungal pathogen to the CNS. Finally, once in the CNS, Cryptococcus spp use substrates such as the neurotransmitters dopamine or adrenaline, which aid in the synthesis of melanin, a cell wall component that enhances protection from oxidative stress and phagocytosis.
对隐球菌病发病机制的大部分了解来源于对新生隐球菌的研究。当吸入孢子或干燥细胞时,隐球菌属建立肺部感染。一旦进入肺,酵母可以到达血流并进入CNS,通过跨细胞迁移穿过血脑屏障的微血管内皮进入CSF。穿过血脑屏障的其他机制包括所谓的特洛伊木马机制,即隐球菌通过隐藏在单核细胞内或通过使用细胞旁迁移途径穿过内皮。提出了三种假说来解释真菌嗜神经性。首先,基底神经节中的神经元底物被认为可促进隐球菌生长和存活。CNS,尤其是血管周围间隙(Virchow-Robin间隙),可能作为隐球菌属的微环境。我们的患者显示了由大脑中动脉穿孔豆纹支供血的实质血管周围间隙内隐球菌属感染的证据。在其他血管周围间隙发现感染的进一步体征,包括丘脑、脑室周围白质、小脑和中脑(图2)。其次,似乎存在可能吸引这种真菌病原体进入CNS的特异性神经元受体。最后,一旦进入CNS,隐球菌属使用神经递质多巴胺或肾上腺素等底物,有助于合成黑色素,黑色素是一种细胞壁成分,可增强对氧化应激和吞噬作用的保护。
The precise mechanism leading to intracranial hypertension in cryptococcal infection remains to be elucidated. Circulation of the CSF is a dynamic process, and its regulation is responsible for cerebral homeostasis. Under regular conditions, CSF is produced in the choroid plexus at a rate of 20 mL/h to reach a volume of about 125–150 mL, of which roughly 20% is contained in the ventricular system and the rest circulates in cranial and spinal subarachnoid spaces. Most CSF is continuously resorbed in the arachnoid villi located in the intracranial venous sinuses, and the rest is resorbed by spinal nerve and some cranial nerve sheaths. The Monro-Kellie hypothesis states that the expansion of one of the intracranial components of the brain, CSF, or intravascular blood, is at the expense of a reduction in the other component. As CSF accumulates in the subarachnoid spaces and the compensatory mechanism becomes exhausted, pliable blood vessels are compressed and cerebral blood flow is reduced. When intracranial pressure reaches 50–60 mm Hg, pressure in the arteries of the circle of Willis becomes compromised, producing global cerebral ischaemia and potentially leading to brain death. Because the cranium is a fixed vault, the increase in the volume produces herniation of brain tissue, causing central and tonsillar herniation, similar to what occurred to our patient.
隐球菌感染导致颅内高压的确切机制仍有待阐明。CSF循环是一个动态过程,其调节负责脑内稳态。在常规条件下,CSF在脉络丛中以20 mL/h的速度产生,达到约125-150 mL的体积,其中约20%包含在脑室系统中,其余在颅和脊髓蛛网膜下腔中循环。大多数CSF在位于颅内静脉窦的蛛网膜绒毛中持续吸收,其余被脊神经和部分颅神经鞘吸收。Monro-Kellie假设指出,脑的颅内成分之一CSF或血管内血液的扩张以牺牲另一种成分的减少为代价。随着CSF在蛛网膜下腔积聚,代偿机制耗尽,柔韧的血管受到压迫,脑血流量减少。当颅内压达到50-60 mmHg时,Willis环动脉内的压力受损,产生全脑缺血,并可能导致脑死亡。由于颅骨是一个固定的穹隆,体积的增加产生脑组织的疝形成,引起中央和扁桃体疝形成,与我们的患者发生的。
The reduced rate of removal of CSF is a central aspect of cryptococcosis of the CNS. A report from 1968 described a patient with longstanding cryptococcal meningitis and a substantial increase in intracranial pressure compared with 11 controls, and in whom artificial CSF was infused. Subsequently, many reports have emphasised the possibility that accumulation of CSF and intracranial hypertension in cryptococcosis arise as secondary features to fungal or capsular blockage of CSF outflow at the level of the arachnoid villi. Cryptococcal polysaccharide molecular weight is high (range 2 × 107 – 2 × 108 Da) and can spread rapidly through the brain, as shown by implantation experiments in rats.45,46 Furthermore, animal models using intracerebral injections of purified cryptococcal polysaccharide caused interstitial oedema. This interstitial oedema may not be as visible as in other conditions, due to the concurrent marked accumulation of CSF. Thus, coating of the brain parenchyma by large cryptococcal polysaccharides has been suggested to result in a so-called frozen brain with decreased compliance that leaves ventricles unable to accommodate additional CSF that is inadequately resorbed. This process has been proposed to explain the frequent lack of enlargement of ventricles identified in some patients with either C neoformans or C gattii infection, despite the documented presence of elevated intracranial pressure. An alternative explanation for this process is that, in view of the frequent subacute clinical nature of cryptococcal CNS infection, the distribution of high quantities of the fungus and its polysaccharide in the CNS might allow the gradual establishment of pressure equilibrium between the ventricles, the subarachnoid space, and the interstitial space in the parenchyma. The absence of any decompression in the ventricles after the insertion of ventriculoperitoneal shunts confirms this possible mechanism.
CSF清除率降低是CNS隐球菌病的核心方面。1968年的一份报告描述了与11例对照患者相比,1例长期隐球菌性脑膜炎和颅内压显著升高的患者,在该患者中输注了人工CSF。随后,许多报告强调了隐球菌病中CSF蓄积和颅内高压作为蛛网膜绒毛水平CSF流出的真菌或包膜堵塞的次要特征的可能性。隐球菌多糖分子量高(范围2 × 107-2 × 108 Da),可迅速通过大脑扩散,大鼠植入实验显示。此外,使用脑内注射纯化隐球菌多糖的动物模型引起间质性水肿。由于同时存在明显的CSF蓄积,这种间质性水肿可能不如其他疾病中可见。因此,大隐球菌多糖包被脑实质可导致所谓的冷冻脑,顺应性降低,使脑室不能容纳吸收不充分的额外CSF。这一过程被提出来解释在一些新生C细胞或格特C细胞感染患者中发现的脑室经常没有扩大,尽管有文献报道存在颅内压升高。这一过程的另一种解释是,鉴于隐球菌CNS感染的常见亚急性临床性质,CNS中大量真菌及其多糖的分布可能允许脑室、蛛网膜下腔之间逐渐建立压力平衡,和实质中的组织间隙。脑室腹膜分流管插入后脑室内无任何减压证实了这种可能的机制。
We further speculate that the presence of Cryptococci spp, mucinous material, and capsular polysaccharide deposition might facilitate this pressure equilibrium, given that the CSF moves freely with interstitial fluid in the brain parenchyma via the Virchow-Robin perivascular spaces. In this sense, cerebral oedema associated with cryptococcal infection is mostly interstitial due to decreased CSF outflow, whereas the cerebral oedema leading to increased intracranial volume in acute bacterial meningitis is a combination of interstitial (decreased outflow caused by meningeal inflammation) and vasogenic (by disruption of the blood–brain barrier by polymorphonuclear infiltration) components. The heavy deposition of capsular cryptococcal polysaccharide antigen in brain tissue might result in persistence of antigen in CSF months or years after completion of adequate antifungal therapy.
我们进一步推测隐球菌属、粘液物质和荚膜多糖沉积的存在可能促进这种压力平衡,因为CSF通过Virchow-Robin血管周围间隙与脑实质中的间质液自由移动。从这个意义上说,由于CSF流出减少,与隐球菌感染相关的脑水肿多为间质性,然而,急性细菌性脑膜炎中导致颅内体积增加的脑水肿是间质(脑膜炎症引起的流出减少)和血管源性(多形核细胞浸润破坏血脑屏障)成分的组合。荚膜隐球菌多糖抗原在脑组织中的大量沉积可能导致在完成充分的抗真菌治疗后数月或数年CSF中抗原持续存在。
Management of intracranial hypertension
Treatment guidelines for cryptococcosis have historically distinguished therapeutic approaches based on the presence or absence of HIV infection and whether treating pulmonary or CNS disease. Although treatment guidelines have historically lacked specific recommendations for management of severe C gattii disease, recent experiences illustrate the need for consideration of more aggressive management of CNS inflammatory complications, including procedures to address intracranial hypertension and, in some cases, early use of dexamethasone.
隐球菌病的治疗指南历来根据是否存在HIV感染以及是否治疗肺部或CNS疾病来区分治疗方法。尽管历史上治疗指南缺乏管理重度格特隐球菌疾病的具体建议,但最近的经验表明,需要考虑更积极地管理CNS炎性并发症,包括解决颅内高压的程序,以及在某些情况下早期使用地塞米松。
Intracranial hypertension is associated with poor clinical outcomes, and it is often a potentially remediable component of cryptococcal disease. Thus, prevention of secondary brain damage from intracranial hypertension is a central focus in management of cryptococcal meningitis, with serial lumbar punctures needed as an adjunct to antifungal therapy. Daily lumbar puncture is currently recommended to relieve intracranial pressure until pressure is below 25 cm of water and symptoms (ie, headache, seizures, improved cognition, or improvement of any focal sign) are stabilised for two consecutive days. If pressure remains raised, lumbar percutaneous drains or ventriculoperitoneal shunts are currently recommended. Our experience is that ventriculostomy should also be considered in severe cases of C gattii meningoencephalitis to allow for removal of more CSF than can be safely removed by daily lumbar puncture. Ventriculoperitoneal shunt placement should also be considered in people who have uncontrollable intracranial hypertension, even in the setting of normal ventricular size.
颅内高压与临床结局较差相关,并且通常是隐球菌疾病的潜在可治疗组分。因此,预防颅内高压引起的继发性脑损伤是隐球菌性脑膜炎治疗的中心焦点,需要连续腰椎穿刺作为抗真菌治疗的辅助治疗。目前建议每日进行腰椎穿刺以缓解颅内压,直至压力低于25 cmH20且症状(即头痛、癫痫发作、认知改善或任何局灶性体征改善)连续两天稳定。如果压力仍然升高,目前建议进行腰椎经皮引流或脑室腹膜分流术。我们的经验是,在格特C脑膜脑炎的严重病例中也应考虑脑室造口术,以允许清除比每日腰椎穿刺可安全清除更多的CSF。在患有无法控制的颅内高压的患者中,也应考虑放置脑室腹膜分流管,即使是在脑室尺寸正常的情况下。
Steroids are potentially an important adjunct to antifungal therapy in cases of C neoformans infection with suspected IRIS, although randomised trials have not proven their benefit. Dexamethasone is postulated to inhibit the secretion of cryptococcal glucuro- noxylomannan-induced vascular endothelial growth factor A by mononuclear cells, which can cause damage to the blood–brain barrier. Dexamethasone can thus reduce meningeal inflammation caused by Cryptococcus spp and decrease sterile arachnoiditis during episodes of IRIS. A retrospective study of 16 patients with C gattii infection of the CNS from Papua New Guinea reported that those who received steroids had decreased rates of vision loss and blindness, with improvement in vision in three patients. Other studies have reported substantial clinical improvement in patients with severe cases of meningoencephalitis receiving dexamethasone. However, our patient’s neurological status was rapidly deteriorating by the time we initiated the use of dexamethasone, and this intervention probably took place too far into her clinical course to provide any substantial benefit. Therefore, although more clinical studies are necessary to define optimal treatment guidelines, we suggest that on the basis of existing literature, steroids be considered for patients with intracranial hypertension caused by C gattii infection, particularly in severe cases of meningoencephalitis, and for patients with persistent or worsening symptoms, despite improvements in parameters of fungal burden (antigen titres and CSF sterility).
在疑似IRIS的新型隐球菌感染病例中,类固醇可能是抗真菌治疗的重要辅助治疗,尽管随机试验尚未证实其获益。据推测,地塞米松可抑制单核细胞分泌隐球菌葡萄糖-诺木甘露聚糖诱导的血管内皮生长因子A,这可能导致血脑屏障损伤。因此,地塞米松可以减少隐球菌引起的脑膜炎症,并减少IRIS发作期间的无菌性蛛网膜炎。一项对来自巴布亚新几内亚的16例CNS格特隐球菌感染患者的回顾性研究报告,接受类固醇治疗的患者视力丧失和失明率降低,3例患者视力改善。其他研究报告,接受地塞米松治疗的脑膜脑炎重度病例患者的临床显著改善。然而,当我们开始使用地塞米松时,我们的患者的神经功能状态迅速恶化,这种干预可能发生在她的临床病程太远而无法提供任何实质性获益。因此,尽管需要更多的临床研究来确定最佳的治疗指南,但我们建议在现有文献的基础上,对于格特隐球菌感染引起的颅内高压患者,特别是脑膜脑炎的严重病例,以及症状持续或恶化的患者,应考虑使用类固醇,尽管真菌负荷参数(抗原滴度和CSF无菌性)有所改善。
Antifungal therapy
Microbial control in CNS disease caused by C gattii can often necessitate prolonged courses of amphotericin formulations (plus flucytosine). A recent report from Australia suggested that improved outcomes among those with CNS disease were associated with a 6-week course of amphotericin and flucytosine (table). Recent epidemio- logical studies demonstrate that some strains of C gattii, especially those of the VGII molecular type, have relatively low susceptibilities to fluconazole, with sustained susceptibility to voriconazole and posaconazole. Although no reported data correlates low susceptibility to fluconazole by minimal inhibitory concentration to clinical outcomes, our experience is that these other azoles might have relatively improved activity when used during the continuation phase of therapy, during which the goal is to maintain oral azole therapy for weeks to months (average 6months) after the patient completed the induction phase of treatment with amphotericin B-based formulations. Therefore, further research is needed to prospectively evaluate treatment outcomes of C gattii infections and minimal inhibitory concentration values with fluconazole.
CNS疾病患者的结果与6周疗程的两性霉素和氟胞嘧啶相关(表)。最近的流行病学研究表明,一些格特C菌株,尤其是VGII分子型菌株,对氟康唑的敏感性相对较低,对伏立康唑和泊沙康唑的敏感性持续存在。尽管没有报告的数据表明最低抑菌浓度对氟康唑的低敏感性与临床结局相关,但我们的经验是,当在继续治疗阶段使用时,这些其他唑类药物的活性可能相对改善,在此期间,目标是在患者完成两性霉素B制剂的诱导治疗期后维持口服唑类药物治疗数周至数月(平均6个月)。因此,需要进一步的研究来前瞻性评价格特隐球菌感染的治疗结局和氟康唑的最小抑菌浓度值。
Conclusion
Our case illustrates an unfortunate and dramatic clinical outcome of severe infection caused by this emerging fungal pathogen. While we await further research to guide the optimum care of C gattii meningoencephalitis, we suggest that our experiences support a therapeutic approach that considers aggressive CSF drainage to manage intracranial hypertension with percutaneous lumbar drain or ventriculostomy placement early in the course of the disease. Some cases need placement of ventriculoperitoneal shunts. The concomitant use of dexamethasone to decrease (or modulate) a potentially harmful inflammatory response might be an important consideration, although we lack definitive clinical data documenting utility or defining an optimum drug, dose, or timing. Finally, prolonged courses of amphotericin-B- based regimens with flucytosine appear to be needed. Despite the lack of randomised trials, growing clinical experience as well as epidemiological and microbiological data support the use of expanded-spectrum azoles during the continuation phase of treatment.
我们的病例说明了这种新出现的真菌病原体引起的严重感染的不幸和戏剧性的临床结果。当我们等待进一步的研究以指导格特C脑膜脑炎的最佳护理时,我们建议我们的经验支持一种治疗方法,即在疾病早期考虑使用经皮腰椎引流或脑室引流进行积极的CSF引流以治疗颅内高压。有些病例需要放置脑室腹膜分流管。合并使用地塞米松以减少(或调节)潜在有害的炎症反应可能是一个重要的考虑因素,尽管我们缺乏记录效用或确定最佳药物、剂量或时间的明确临床数据。最后,似乎需要延长以两性霉素B为基础的氟胞嘧啶治疗方案的疗程。尽管缺乏随机试验,但越来越多的临床经验以及流行病学和微生物学数据支持在继续治疗阶段使用广谱唑类药物。
原创文章(本站视频密码:66668888),作者:xujunzju,如若转载,请注明出处:https://zyicu.cn/?p=10809

 微信扫一扫
微信扫一扫  支付宝扫一扫
支付宝扫一扫 

![[NEJM临床医学影像]:皮肌炎的眶周水肿](https://zyicu.cn/wordpress/wp-content/uploads/2023/05/image-49-480x300.png)
