Abstract
Importance
Coronavirus disease 2019 (COVID-19) is associated with severe lung damage. Corticosteroids are a possible therapeutic option.
2019年冠状病毒疾病(COVID-19)与重度肺损伤相关。皮质类固醇是一种可能的治疗选择。
Objective
To determine the effect of hydrocortisone on treatment failure on day 21 in critically ill patients with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection and acute respiratory failure.
确定氢化可的松对严重急性呼吸综合征冠状病毒2(SARS-CoV-2)感染和急性呼吸衰竭重症患者第21天治疗失败的影响。
Design, Setting, and Participants
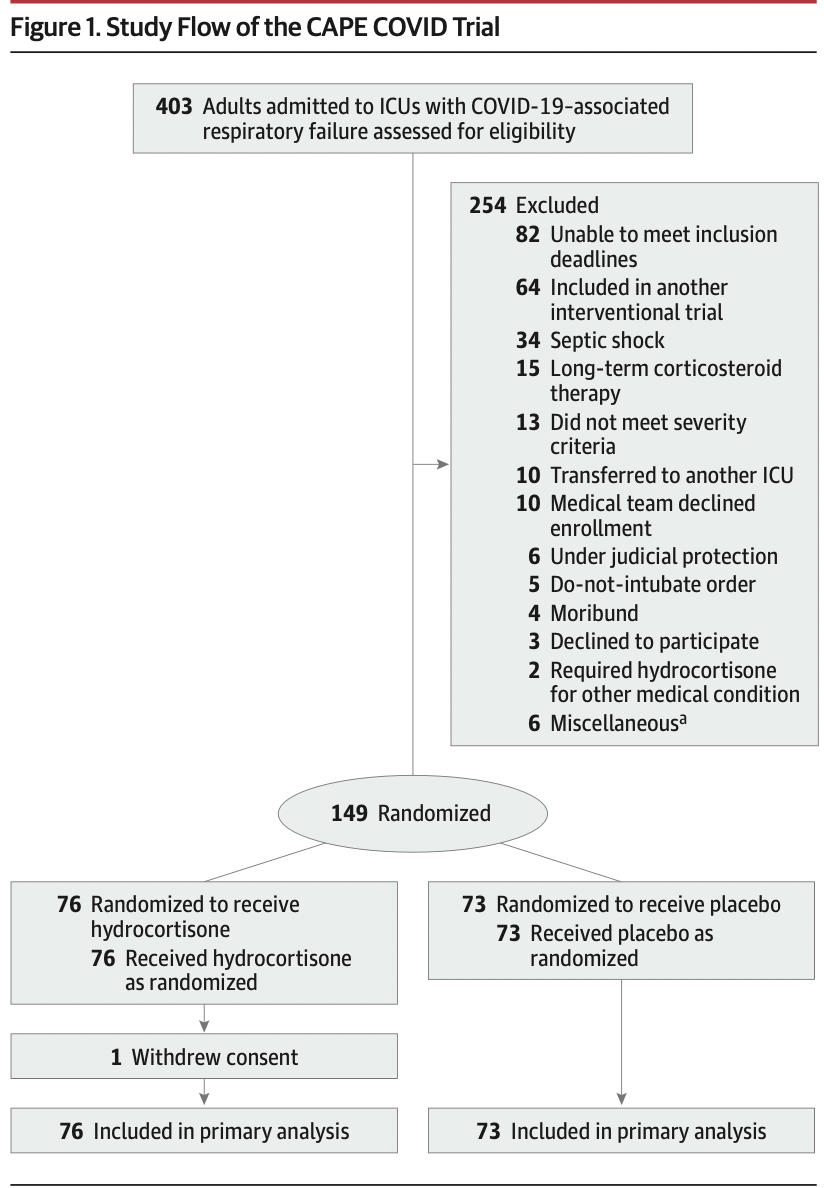
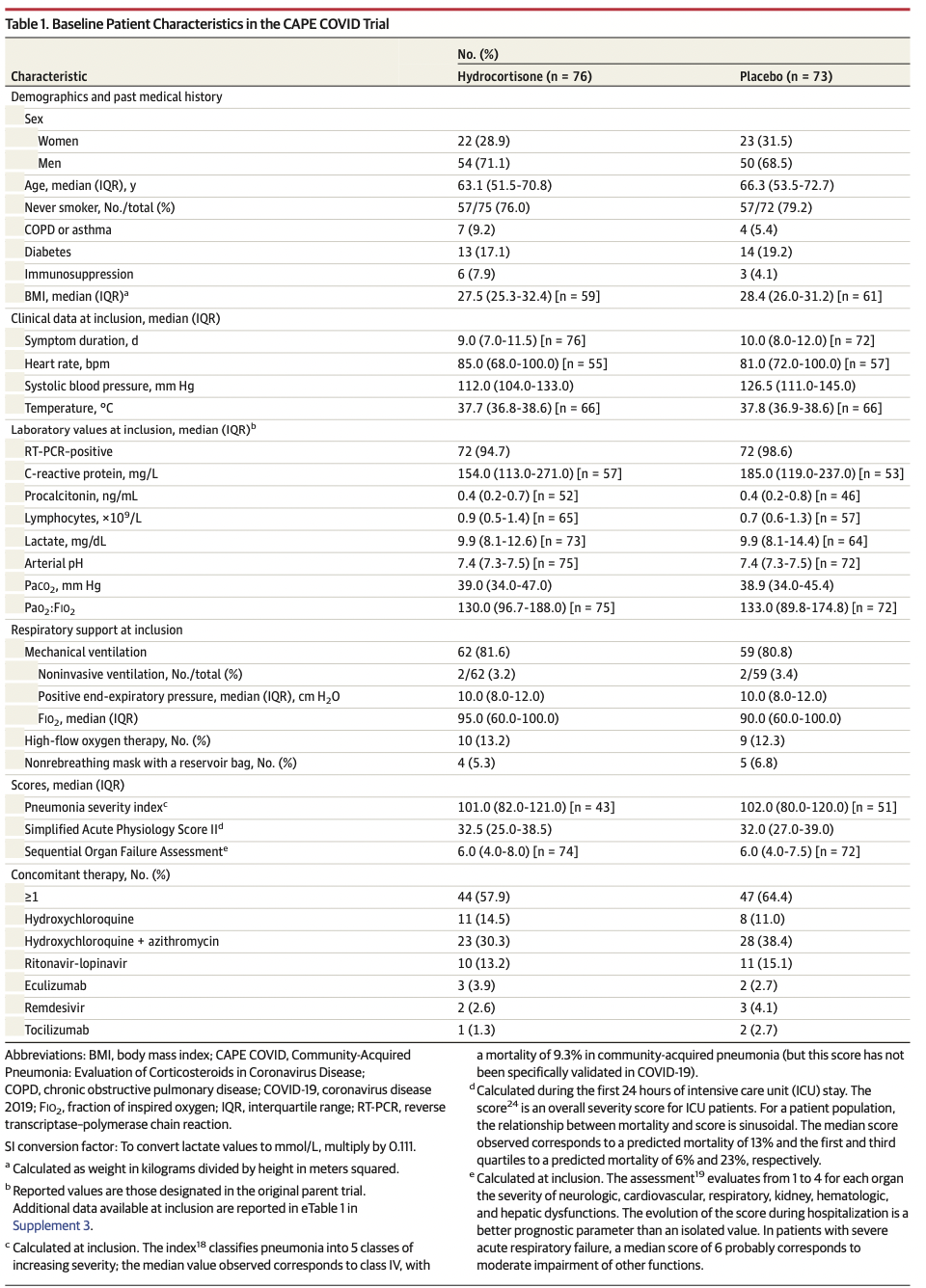
Multicenter randomized double-blind sequential trial conducted in France, with interim analyses planned every 50 patients. Patients admitted to the intensive care unit (ICU) for COVID-19–related acute respiratory failure were enrolled from March 7 to June 1, 2020, with last follow-up on June 29, 2020. The study intended to enroll 290 patients but was stopped early following the recommendation of the data and safety monitoring board.
在法国进行的多中心随机双盲序贯试验,计划每50例患者进行中期分析。2020年3月7日至6月1日入重症监护室(ICU)治疗COVID-19相关急性呼衰的患者,末次随访日期为2020年6月29日。本研究计划入组290例患者,但根据数据和安全性监测委员会的建议提前停止。
Interventions
Patients were randomized to receive low-dose hydrocortisone (n = 76) or placebo (n = 73).
患者随机接受低剂量氢化可的松(n = 76)或安慰剂(n = 73)。
Main Outcomes and Measures
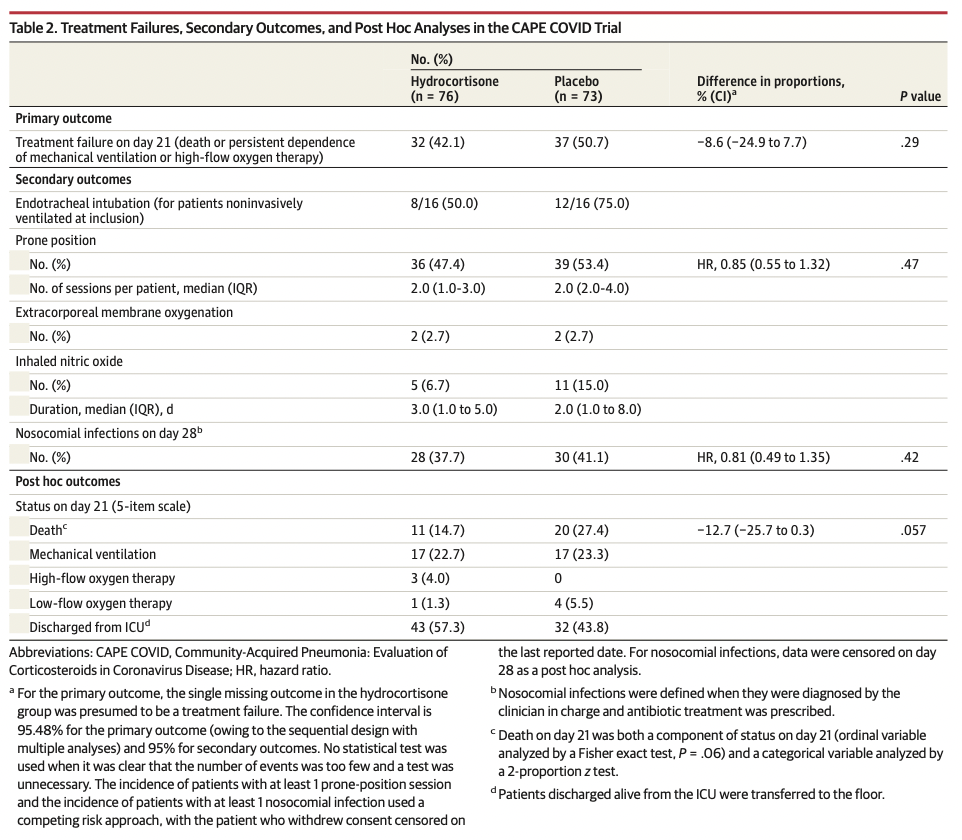
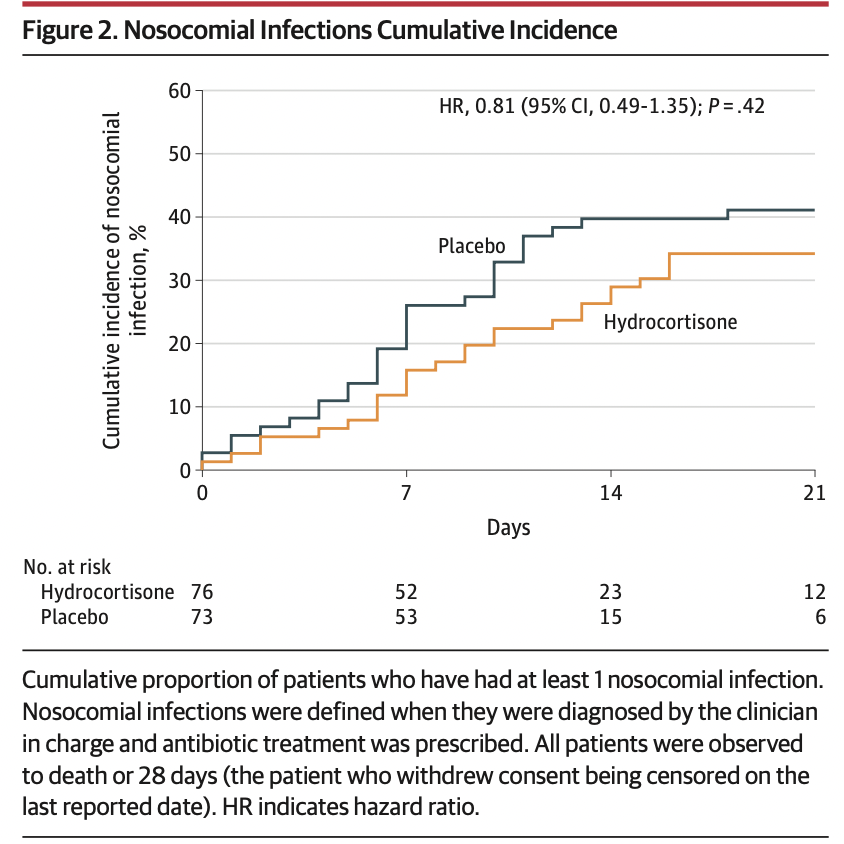
The primary outcome, treatment failure on day 21, was defined as death or persistent dependency on mechanical ventilation or high-flow oxygen therapy. Prespecified secondary outcomes included the need for tracheal intubation (among patients not intubated at baseline); cumulative incidences (until day 21) of prone position sessions, extracorporeal membrane oxygenation, and inhaled nitric oxide; Pao2:Fio2 ratio measured daily from day 1 to day 7, then on days 14 and 21; and the proportion of patients with secondary infections during their ICU stay.
主要结局,第21天治疗失败,定义为死亡或持续依赖机械通气或高流量氧疗。预先指定的次要结果包括气管插管的需要(在基线时未插管的患者中);俯卧位、体外膜肺氧合和吸入一氧化氮的累积发生率(直到第21天);从第1天到第7天每天测量PaO2:FiO2比值,然后在第14天和第21天测量;以及在ICU住院期间继发感染的患者比例。
Results
The study was stopped after 149 patients (mean age, 62.2 years; 30.2% women; 81.2% mechanically ventilated) were enrolled. One hundred forty-eight patients (99.3%) completed the study, and there were 69 treatment failure events, including 11 deaths in the hydrocortisone group and 20 deaths in the placebo group. The primary outcome, treatment failure on day 21, occurred in 32 of 76 patients (42.1%) in the hydrocortisone group compared with 37 of 73 (50.7%) in the placebo group (difference of proportions, –8.6% [95.48% CI, –24.9% to 7.7%]; P = .29). Of the 4 prespecified secondary outcomes, none showed a significant difference. No serious adverse events were related to the study treatment.
在入组149例患者(平均年龄,62.2岁;30.2%为女性;81.2%为机械通气)后停止研究。148例患者(99.3%)完成研究,发生69例治疗失败事件,包括氢化可的松组11例死亡和安慰剂组20例死亡。氢化可的松组76例患者中有32例(42.1%)在第21天治疗失败,而安慰剂组73例患者中有37例(50.7%)在第21天治疗失败(比例差异,-8.6%[95.48%CI,-24.9%-7.7%];P = .29).在预先规定的4个次要结局中,没有一个显示出显著差异。无与研究治疗相关的严重不良事件。
Conclusions and Relevance
In this study of critically ill patients with COVID-19 and acute respiratory failure, low-dose hydrocortisone, compared with placebo, did not significantly reduce treatment failure (defined as death or persistent respiratory support) at day 21. However, the study was stopped early and likely was underpowered to find a statistically and clinically important difference in the primary outcome.
在这项COVID-19和急性呼吸衰竭重症患者的研究中,与安慰剂相比,低剂量氢化可的松未显著降低第21天的治疗失败(定义为死亡或持续呼吸支持)。但是,该研究提前停止,可能不足以发现主要结局的统计学和临床重要差异。
Trial Registration
ClinicalTrials.gov Identifier: NCT02517489
治疗组的氢化可的松方案如下:初始剂量为200 mg/日,D1-D7;100 mg/日,D8-D11; 50mg/日,D12-D14;共14天。如果患者的呼吸和一般状况在第4天充分改善,则使用短期治疗方案(200 mg/日治疗4天,然后100 mg/日治疗2天,然后50 mg/日治疗2天,共治疗8天。
原创文章(本站视频密码:66668888),作者:xujunzju,如若转载,请注明出处:https://zyicu.cn/?p=9848

 微信扫一扫
微信扫一扫  支付宝扫一扫
支付宝扫一扫