Abstract
Background: Whether the antiinflammatory and immunomodulatory effects of glucocorticoids may decrease mortality among patients with severe community-acquired pneumonia is unclear.
背景:糖皮质激素的抗炎和免疫调节作用是否可以降低重症社区获得性肺炎患者的死亡率尚不清楚。
Methods: In this phase 3, multicenter, double-blind, randomized, controlled trial, we assigned adults who had been admitted to the intensive care unit (ICU) for severe community-acquired pneumonia to receive intravenous hydrocortisone (200 mg daily for either 4 or 8 days as determined by clinical improvement, followed by tapering for a total of 8 or 14 days) or to receive placebo. All the patients received standard therapy, including antibiotics and supportive care. The primary outcome was death at 28 days.
方法:在这项3期、多中心、双盲、随机、对照试验中,我们将因重症社区获得性肺炎被收入重症监护病房(ICU)的成人患者随机分组,一组接受氢化可的松静脉给药(每日200 mg,根据临床改善情况治疗4日或8日,之后减量共8日或14日),另一组接受安慰剂。所有患者均接受标准治疗,包括抗生素和支持治疗。主要结局是28日时的死亡。
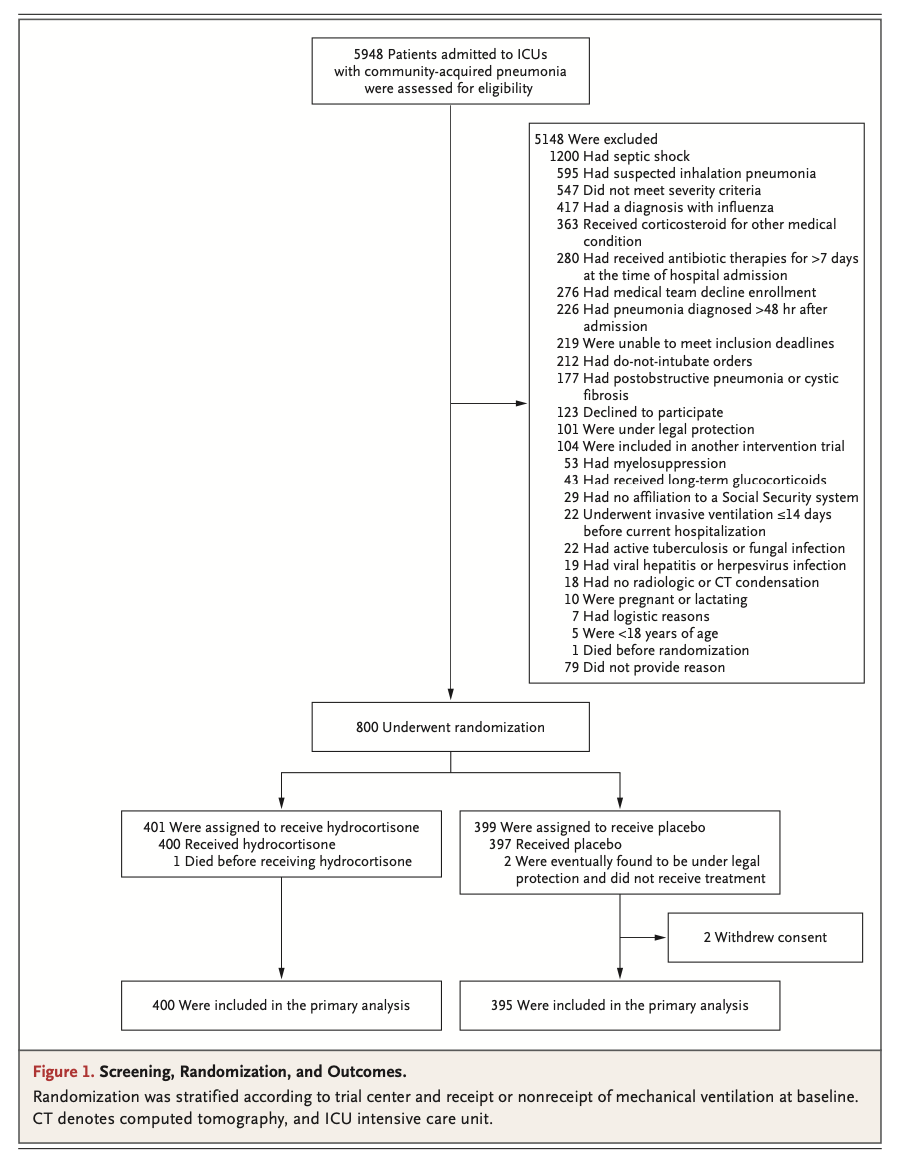
Results: A total of 800 patients had undergone randomization when the trial was stopped after the second planned interim analysis. Data from 795 patients were analyzed. By day 28, death had occurred in 25 of 400 patients (6.2%; 95% confidence interval [CI], 3.9 to 8.6) in the hydrocortisone group and in 47 of 395 patients (11.9%; 95% CI, 8.7 to 15.1) in the placebo group (absolute difference, -5.6 percentage points; 95% CI, -9.6 to -1.7; P = 0.006). Among the patients who were not undergoing mechanical ventilation at baseline, endotracheal intubation was performed in 40 of 222 (18.0%) in the hydrocortisone group and in 65 of 220 (29.5%) in the placebo group (hazard ratio, 0.59; 95% CI, 0.40 to 0.86). Among the patients who were not receiving vasopressors at baseline, such therapy was initiated by day 28 in 55 of 359 (15.3%) of the hydrocortisone group and in 86 of 344 (25.0%) in the placebo group (hazard ratio, 0.59; 95% CI, 0.43 to 0.82). The frequencies of hospital-acquired infections and gastrointestinal bleeding were similar in the two groups; patients in the hydrocortisone group received higher daily doses of insulin during the first week of treatment.
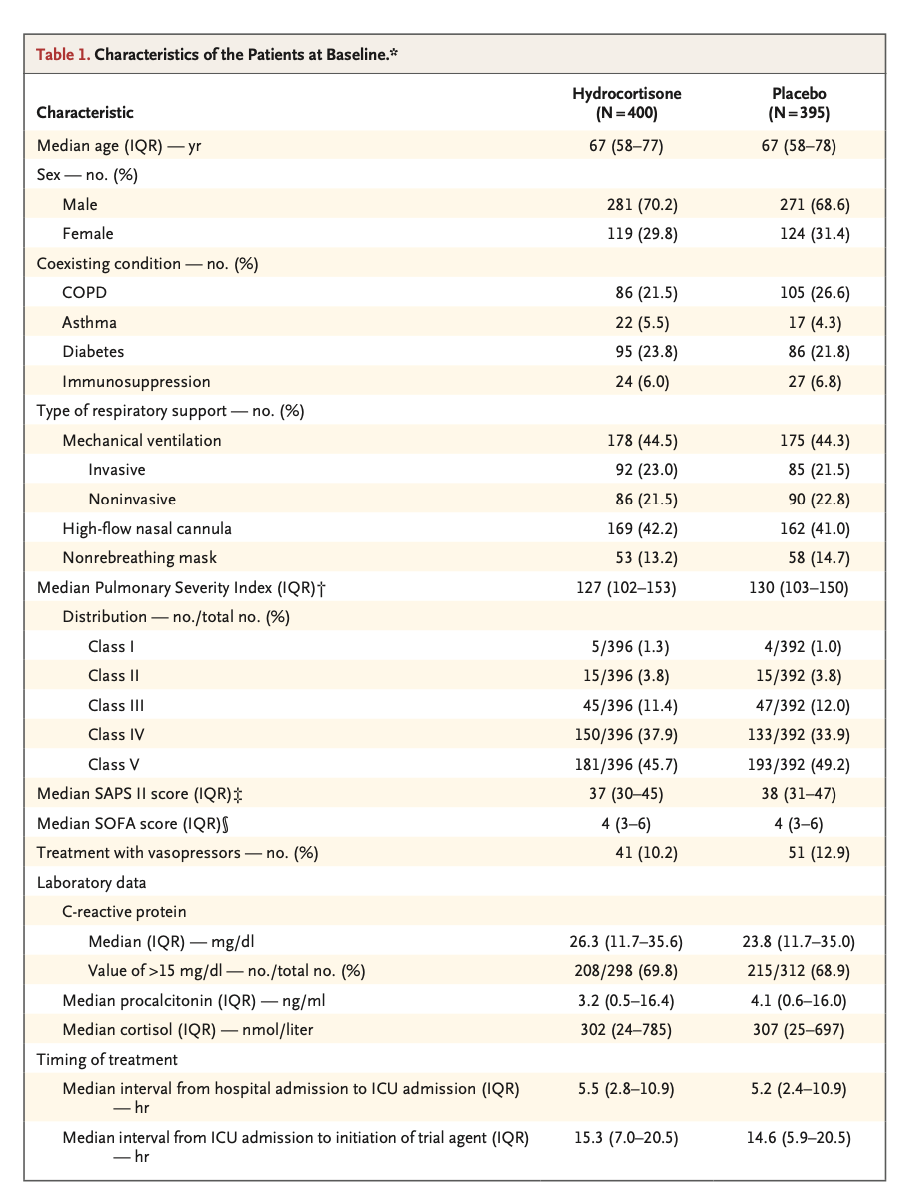
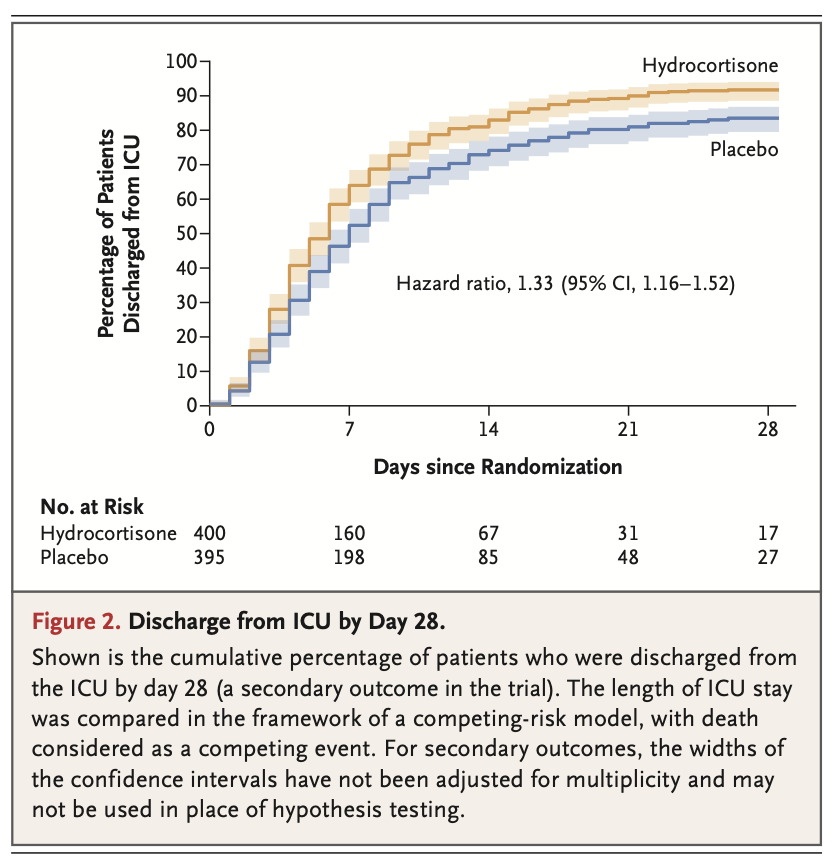
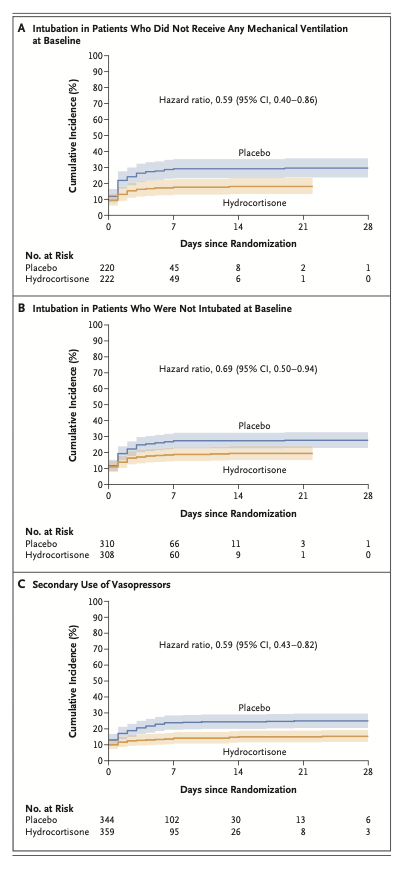
结果:在第二次计划的中期分析结束后试验终止时,共有800例患者接受了随机分组。共纳入795例患者。截至第28日,400例患者中有25例死亡(6.2%;95%置信区间[CI], 3.9 ~ 8.6)和395例患者中的47例(11.9%;95% CI, 8.7 ~ 15.1)(绝对差异,-5.6个百分点;95% CI: -9.6 ~ -1.7;P = 0.006)。在基线时未接受机械通气的患者中,氢化可的松组222例患者中的40例(18.0%)和安慰剂组220例患者中的65例(29.5%)接受了气管插管(风险比,0.59;95% ci, 0.59 ~ 0.59)(表2)。95% CI, 0.40 ~ 0.86)。在基线时未接受血管升压药治疗的患者中,从第28日开始,氢化可的松组359例患者中的55例(15.3%)和安慰剂组344例患者中的86例(25.0%)开始了此类治疗(风险比,0.59;95% ci, 0.59 ~ 0.59)。95% CI, 0.43 ~ 0.82)。两组的医院获得性感染和消化道出血发生率相似;氢化可的松组患者在第1周治疗期间接受了较高的每日胰岛素剂量。
Conclusions: Among patients with severe community-acquired pneumonia being treated in the ICU, those who received hydrocortisone had a lower risk of death by day 28 than those who received placebo. (Funded by the French Ministry of Health; CAPE COD ClinicalTrials.gov number, NCT02517489.).
结论:在ICU接受治疗的重症社区获得性肺炎患者中,与接受安慰剂治疗的患者相比,接受氢化可的松治疗的患者截至第28日时的死亡风险较低。(由法国卫生部资助;CAPE COD在ClinicalTrials.gov注册号为NCT02517489)。
原创文章(本站视频密码:66668888),作者:xujunzju,如若转载,请注明出处:https://zyicu.cn/?p=15692

 微信扫一扫
微信扫一扫  支付宝扫一扫
支付宝扫一扫