Abstract
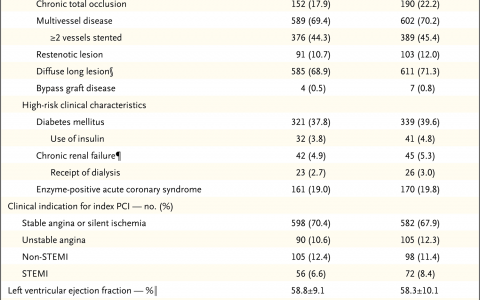
Background: In patients with coronary artery disease who are being evaluated for percutaneous coronary intervention (PCI), procedures can be guided by fractional flow reserve (FFR) or intravascular ultrasonography (IVUS) for decision making regarding revascularization and stent implantation. However, the differences in clinical outcomes when only one method is used for both purposes are unclear.
在接受经皮冠状动脉介入治疗(PCI)评估的冠心病患者中,可以通过血流储备分数(FFR)或血管内超声成像(IVUS)来指导有关血运重建和支架植入的决策。然而,当一种方法同时用于两种目的时,临床结局的差异尚不明确。
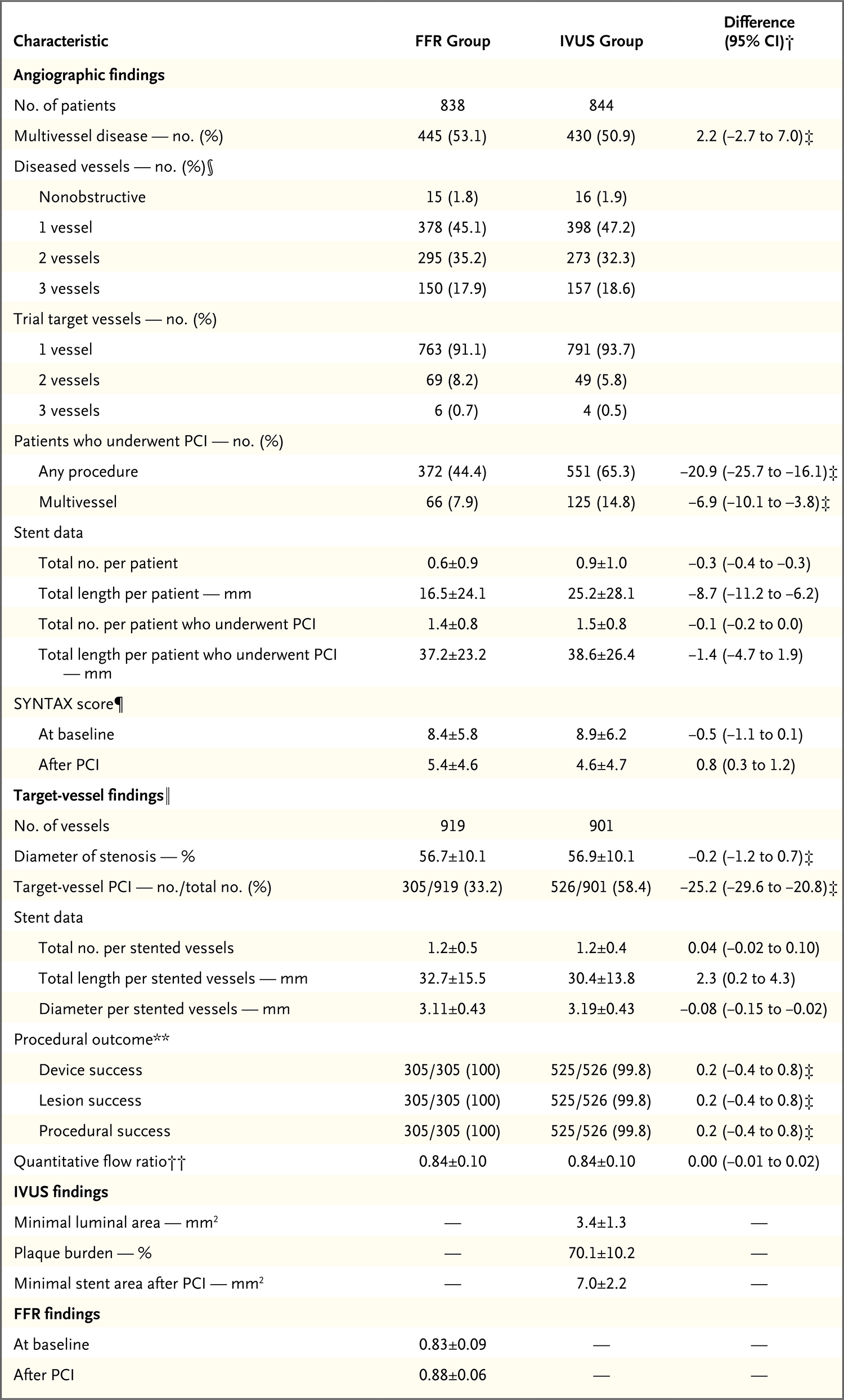
Methods: We randomly assigned 1682 patients who were being evaluated for PCI for the treatment of intermediate stenosis (40 to 70% occlusion by visual estimation on coronary angiography) in a 1:1 ratio to undergo either an FFR-guided or IVUS-guided procedure. FFR or IVUS was to be used to determine whether to perform PCI and to assess PCI success. In the FFR group, PCI was to be performed if the FFR was 0.80 or less. In the IVUS group, the criteria for PCI were a minimal lumen area measuring either 3 mm2 or less or measuring 3 to 4 mm2 with a plaque burden of more than 70%. The primary outcome was a composite of death, myocardial infarction, or revascularization at 24 months after randomization. We tested the noninferiority of the FFR group as compared with the IVUS group (noninferiority margin, 2.5 percentage points).
我们以1∶1的比例将因中度狭窄(冠状动脉造影目测40%~70%堵塞)接受PCI治疗评估的1,682例患者随机分组,分别接受FFR指导PCI或IVUS指导PCI。FFR或IVUS用于确定是否进行PCI并评估PCI成功与否。在FFR组中,如果FFR≤0.80,则进行PCI。在IVUS组中,PCI标准是最小管腔面积≤3 mm2或最小管腔面积为3~4 mm2并且斑块负荷>70%。主要结局是随机分组后24个月时由死亡、心肌梗死或血运重建构成的复合结局。我们检验了FFR组与IVUS组相比的非劣效性(非劣效性界值,2.5个百分点)。
Results: The frequency of PCI was 44.4% among patients in the FFR group and 65.3% among those in the IVUS group. At 24 months, a primary-outcome event had occurred in 8.1% of the patients in the FFR group and in 8.5% of those in the IVUS group (absolute difference, -0.4 percentage points; upper boundary of the one-sided 97.5% confidence interval, 2.2 percentage points; P = 0.01 for noninferiority). Patient-reported outcomes as reported on the Seattle Angina Questionnaire were similar in the two groups.
在FFR组和IVUS组中,接受PCI的患者比例分别为44.4%和65.3%。24个月时,FFR组8.1%的患者和IVUS组8.5%的患者发生了主要结局事件(绝对差,-0.4个百分点;单侧97.5%置信区间上限,2.2个百分点;非劣效性的P=0.01)。两组西雅图心绞痛问卷上患者报告的结局相似。

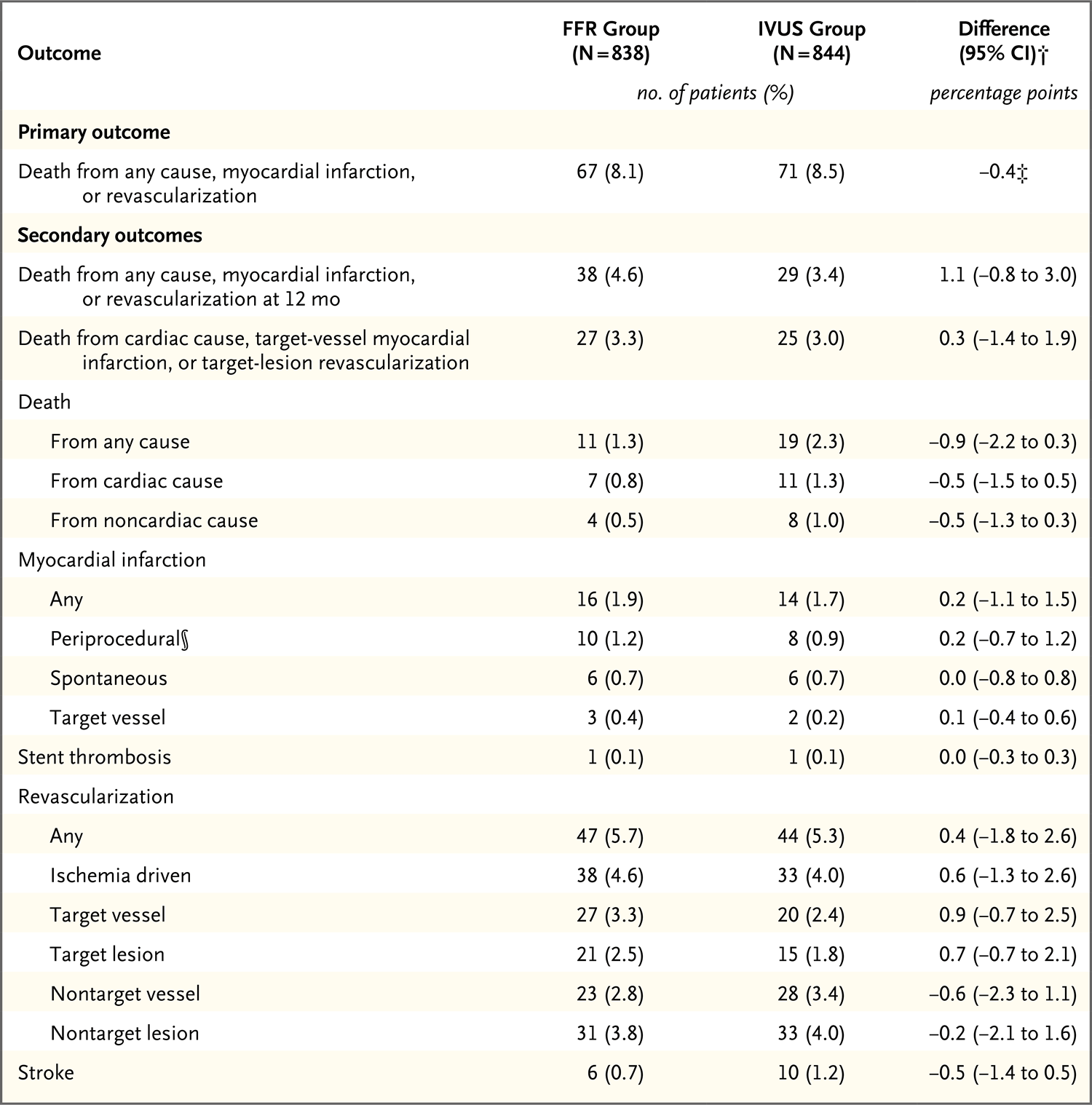
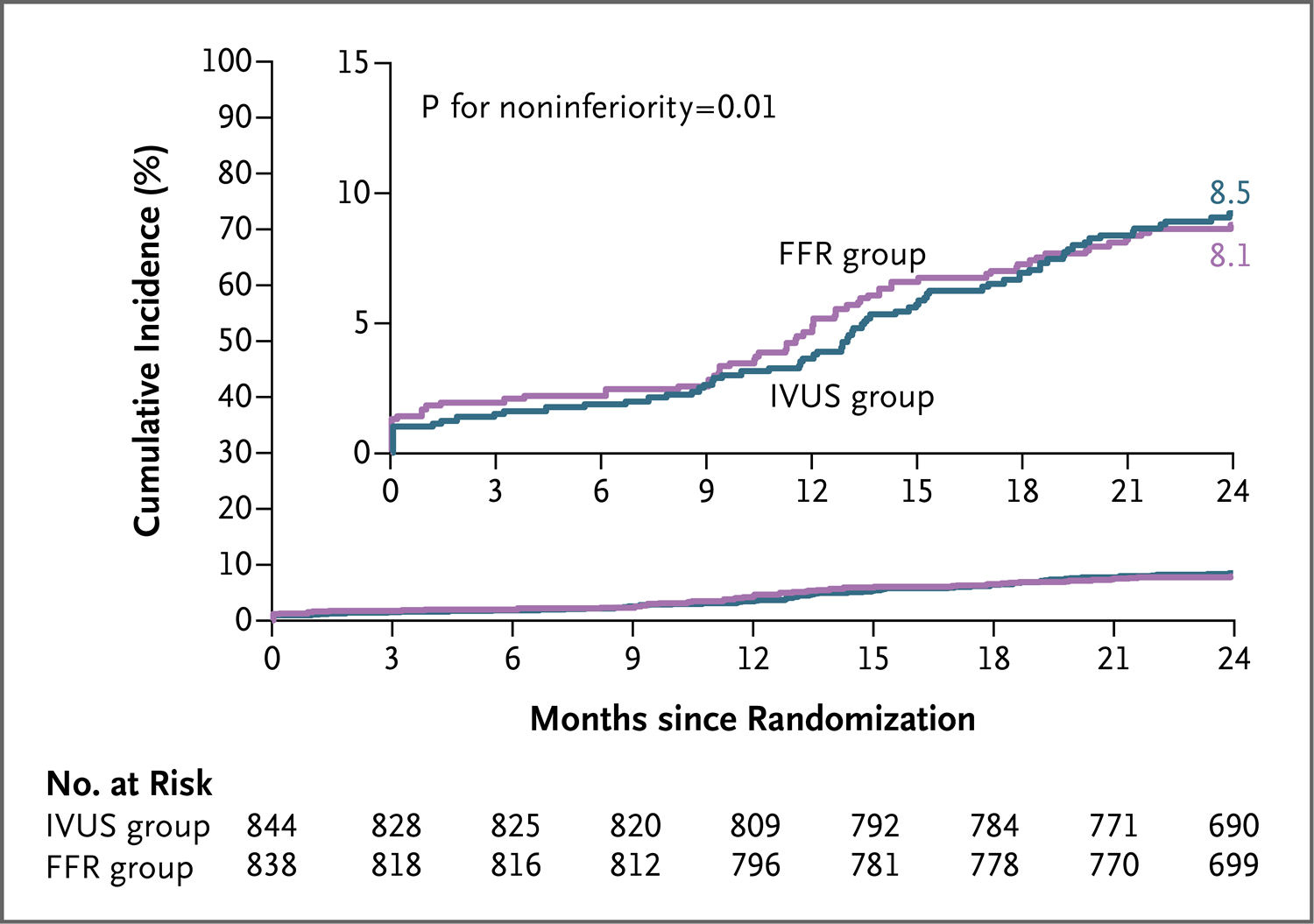
Conclusions: In patients with intermediate stenosis who were being evaluated for PCI, FFR guidance was noninferior to IVUS guidance with respect to the composite primary outcome of death, myocardial infarction, or revascularization at 24 months. (Funded by Boston Scientific; FLAVOUR ClinicalTrials.gov number, NCT02673424.).
在接受PCI评估的冠状动脉中度狭窄患者中,在24个月时的死亡、心肌梗死或血运重建构成的复合主要结局方面,FFR指导PCI不劣于IVUS指导PCI(由波士顿科学公司[Boston Scientific]资助;FLAVOUR在ClinicalTrials.gov注册号为NCT02673424)。
特别申明:本文为转载文章,转载自NEJM,不代表贪吃的夜猫子立场,如若转载,请注明出处:https://www.nejmqianyan.cn/article-info?permalinks=YXQYoa2201546

 微信扫一扫
微信扫一扫  支付宝扫一扫
支付宝扫一扫